Luteoloside Acts as 3C Protease Inhibitor of Enterovirus 71 In Vitro
- PMID: 26870944
- PMCID: PMC4752227
- DOI: 10.1371/journal.pone.0148693
Luteoloside Acts as 3C Protease Inhibitor of Enterovirus 71 In Vitro
Abstract
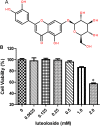
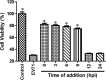
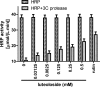
Luteoloside is a member of the flavonoids family that exhibits several bioactivities including anti-microbial and anti-cancer activities. However, the antiviral activity of luteoloside against enterovirus 71 (EV71) and the potential mechanism(s) responsible for this effect remain unknown. In this study, the antiviral potency of luteoloside against EV71 and its inhibitory effects on 3C protease activity were evaluated. First, we investigated the cytotoxicity of luteoloside against rhabdomyosarcoma (RD) cells, which was the cell line selected for an in vitro infection model. In a subsequent antiviral assay, the cytopathic effect of EV71 was significantly and dose-dependently relieved by the administration of luteoloside (EC50 = 0.43 mM, selection index = 5.3). Using a plaque reduction assay, we administered luteoloside at various time points and found that the compound reduced EV71 viability in RD cells rather than increasing defensive mobilization or viral absorption. Moreover, biochemical studies focused on VP1 (a key structural protein of EV71) mRNA transcript and protein levels also revealed the inhibitory effects of luteoloside on the EV71 viral yield. Finally, we performed inhibition assays using luteoloside to evaluate its effect on recombinant 3C protease activity. Our results demonstrated that luteoloside blocked 3C protease enzymatic activity in a dose-dependent manner (IC50 = 0.36 mM) that was similar to the effect of rutin, which is a well-known C3 protease inhibitor. Collectively, the results from this study indicate that luteoloside can block 3C protease activity and subsequently inhibit EV71 production in vitro.
Conflict of interest statement
Figures
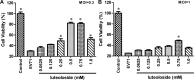


References
-
- Zhou W, Tam KY, Meng M, Shan J, Wang S, Ju W, et al. (2015) Pharmacokinetics screening for multi-components absorbed in the rat plasma after oral administration of traditional Chinese medicine Flos Lonicerae Japonicae-Fructus Forsythiae herb couple by sequential negative and positive ionization ultra-high-performance liquid chromatography/tandem triple quadrupole mass spectrometric detection. J Chromatogr A 1376: 84–97. 10.1016/j.chroma.2014.12.018 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

