Retinal pigment epithelial cell multinucleation in the aging eye - a mechanism to repair damage and maintain homoeostasis
- PMID: 26875723
- PMCID: PMC4854907
- DOI: 10.1111/acel.12447
Retinal pigment epithelial cell multinucleation in the aging eye - a mechanism to repair damage and maintain homoeostasis
Abstract
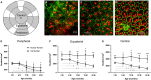
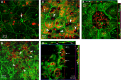
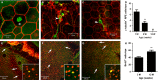
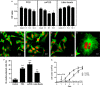
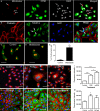
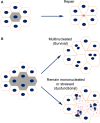
Retinal pigment epithelial (RPE) cells are central to retinal health and homoeostasis. Dysfunction or death of RPE cells underlies many age-related retinal degenerative disorders particularly age-related macular degeneration. During aging RPE cells decline in number, suggesting an age-dependent cell loss. RPE cells are considered to be postmitotic, and how they repair damage during aging remains poorly defined. We show that RPE cells increase in size and become multinucleate during aging in C57BL/6J mice. Multinucleation appeared not to be due to cell fusion, but to incomplete cell division, that is failure of cytokinesis. Interestingly, the phagocytic activity of multinucleate RPE cells was not different from that of mononuclear RPE cells. Furthermore, exposure of RPE cells in vitro to photoreceptor outer segment (POS), particularly oxidized POS, dose-dependently promoted multinucleation and suppressed cell proliferation. Both failure of cytokinesis and suppression of proliferation required contact with POS. Exposure to POS also induced reactive oxygen species and DNA oxidation in RPE cells. We propose that RPE cells have the potential to proliferate in vivo and to repair defects in the monolayer. We further propose that the conventionally accepted 'postmitotic' status of RPE cells is due to a modified form of contact inhibition mediated by POS and that RPE cells are released from this state when contact with POS is lost. This is seen in long-standing rhegmatogenous retinal detachment as overtly proliferating RPE cells (proliferative vitreoretinopathy) and more subtly as multinucleation during normal aging. Age-related oxidative stress may promote failure of cytokinesis and multinucleation in RPE cells.
Keywords: aging; cytokinesis; multinucleation; phagocytosis; photoreceptor outer segments; retinal pigment epithelium.
© 2016 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Figures
Similar articles
-
Mitochondria dynamics in the aged mice eye and the role in the RPE phagocytosis.Exp Eye Res. 2021 Dec;213:108800. doi: 10.1016/j.exer.2021.108800. Epub 2021 Oct 21. Exp Eye Res. 2021. PMID: 34688622
-
Retinal pigment epithelial cells use a MerTK-dependent mechanism to limit the phagocytic particle binding activity of αvβ5 integrin.Biol Cell. 2012 Jun;104(6):326-41. doi: 10.1111/boc.201100076. Epub 2012 Mar 5. Biol Cell. 2012. PMID: 22289110 Free PMC article.
-
Advanced Analysis of Photoreceptor Outer Segment Phagocytosis by RPE Cells in Culture.Methods Mol Biol. 2019;1834:95-108. doi: 10.1007/978-1-4939-8669-9_7. Methods Mol Biol. 2019. PMID: 30324439 Free PMC article.
-
Phagocytosis by the retinal pigment epithelium: New insights into polarized cell mechanics.Bioessays. 2025 Jan;47(1):e2300197. doi: 10.1002/bies.202300197. Epub 2024 Dec 11. Bioessays. 2025. PMID: 39663766 Free PMC article. Review.
-
Role of RPE Phagocytosis in the Retina Metabolic Ecosystem.Adv Exp Med Biol. 2025;1468:429-433. doi: 10.1007/978-3-031-76550-6_70. Adv Exp Med Biol. 2025. PMID: 39930233 Review.
Cited by
-
Autophagy Upregulation by the TFEB Inducer Trehalose Protects against Oxidative Damage and Cell Death Associated with NRF2 Inhibition in Human RPE Cells.Oxid Med Cell Longev. 2020 Jul 21;2020:5296341. doi: 10.1155/2020/5296341. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32774677 Free PMC article.
-
AMPK-induced mitochondrial biogenesis decelerates retinal pigment epithelial cell degeneration under nutrient starvation.BMB Rep. 2023 Feb;56(2):84-89. doi: 10.5483/BMBRep.2022-0125. BMB Rep. 2023. PMID: 36195569 Free PMC article.
-
Effects of senescent secretory phenotype acquisition on human retinal pigment epithelial stem cells.Aging (Albany NY). 2018 Nov 16;10(11):3173-3184. doi: 10.18632/aging.101624. Aging (Albany NY). 2018. PMID: 30444724 Free PMC article.
-
Interaction of High-Molecular Weight Fucoidan from Laminaria hyperborea with Natural Functions of the Retinal Pigment Epithelium.Int J Mol Sci. 2023 Jan 23;24(3):2232. doi: 10.3390/ijms24032232. Int J Mol Sci. 2023. PMID: 36768552 Free PMC article.
-
Autofluorescent Organelles Within the Retinal Pigment Epithelium in Human Donor Eyes With and Without Age-Related Macular Degeneration.Invest Ophthalmol Vis Sci. 2022 Jan 3;63(1):23. doi: 10.1167/iovs.63.1.23. Invest Ophthalmol Vis Sci. 2022. PMID: 35050307 Free PMC article.
References
-
- Abercrombie M (1967) Contact inhibition: the phenomenon and its biological implications. Natl. Cancer Inst. Monogr. 26, 249–277. - PubMed
-
- Anderson JM (2000) Multinucleated giant cells. Curr. Opin. Hematol. 7, 40–47. - PubMed
-
- Aragona M, Panciera T, Manfrin A, Giulitti S, Michielin F, Elvassore N, Dupont S, Piccolo S (2013) A mechanical checkpoint controls multicellular growth through YAP/TAZ regulation by actin‐processing factors. Cell 154, 1047–1059. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

