FLEXITau: Quantifying Post-translational Modifications of Tau Protein in Vitro and in Human Disease
- PMID: 26877193
- PMCID: PMC5808556
- DOI: 10.1021/acs.analchem.5b04509
FLEXITau: Quantifying Post-translational Modifications of Tau Protein in Vitro and in Human Disease
Abstract
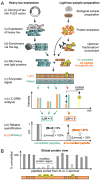
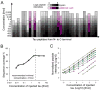
Tauopathies, including Alzheimer's disease (AD), are associated with the aggregation of modified microtubule associated protein tau. This pathological state of tau is often referred to as "hyperphosphorylated". Due to limitations in technology, an accurate quantitative description of this state is lacking. Here, a mass spectrometry-based assay, FLEXITau, is presented to measure phosphorylation stoichiometry and provide an unbiased quantitative view of the tau post-translational modification (PTM) landscape. The power of this assay is demonstrated by measuring the state of hyperphosphorylation from tau in a cellular model for AD pathology, mapping, and calculating site occupancies for over 20 phosphorylations. We further employ FLEXITau to define the tau PTM landscape present in AD post-mortem brain. As shown in this study, the application of this assay provides mechanistic understanding of tau pathology that could lead to novel therapeutics, and we envision its further use in prognostic and diagnostic approaches for tauopathies.
Figures



References
-
- Cripps D, Thomas SN, Jeng Y, Yang F, Davies P, Yang AJ. J Biol Chem. 2006;281:10825–10838. - PubMed
-
- Hanger DP, Byers HL, Wray S, Leung KY, Saxton MJ, Seereeram A, Reynolds CH, Ward MA, Anderton BH. J Biol Chem. 2007;282:23645–23654. - PubMed
-
- Martin L, Latypova X, Terro F. Neurochem Int. 2011;58:458–471. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

