Nociceptin Opioid Receptor (NOP) as a Therapeutic Target: Progress in Translation from Preclinical Research to Clinical Utility
- PMID: 26878436
- PMCID: PMC5001850
- DOI: 10.1021/acs.jmedchem.5b01499
Nociceptin Opioid Receptor (NOP) as a Therapeutic Target: Progress in Translation from Preclinical Research to Clinical Utility
Abstract
In the two decades since the discovery of the nociceptin opioid receptor (NOP) and its ligand, nociceptin/orphaninFQ (N/OFQ), steady progress has been achieved in understanding the pharmacology of this fourth opioid receptor/peptide system, aided by genetic and pharmacologic approaches. This research spawned an explosion of small-molecule NOP receptor ligands from discovery programs in major pharmaceutical companies. NOP agonists have been investigated for their efficacy in preclinical models of anxiety, cough, substance abuse, pain (spinal and peripheral), and urinary incontinence, whereas NOP antagonists have been investigated for treatment of pain, depression, and motor symptoms in Parkinson's disease. Translation of preclinical findings into the clinic is guided by PET and receptor occupancy studies, particularly for NOP antagonists. Recent progress in preclinical NOP research suggests that NOP agonists may have clinical utility for pain treatment and substance abuse pharmacotherapy. This review discusses the progress toward validating the NOP-N/OFQ system as a therapeutic target.
Figures

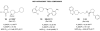

Similar articles
-
The biology of Nociceptin/Orphanin FQ (N/OFQ) related to obesity, stress, anxiety, mood, and drug dependence.Pharmacol Ther. 2014 Mar;141(3):283-99. doi: 10.1016/j.pharmthera.2013.10.011. Epub 2013 Nov 1. Pharmacol Ther. 2014. PMID: 24189487 Free PMC article. Review.
-
Differential In Vitro Pharmacological Profiles of Structurally Diverse Nociceptin Receptor Agonists in Activating G Protein and Beta-Arrestin Signaling at the Human Nociceptin Opioid Receptor.Mol Pharmacol. 2021 Jul;100(1):7-18. doi: 10.1124/molpharm.120.000076. Epub 2021 May 6. Mol Pharmacol. 2021. PMID: 33958480 Free PMC article.
-
The therapeutic potential of nociceptin/orphanin FQ receptor agonists as analgesics without abuse liability.ACS Chem Neurosci. 2013 Feb 20;4(2):214-24. doi: 10.1021/cn300124f. Epub 2012 Nov 6. ACS Chem Neurosci. 2013. PMID: 23421672 Free PMC article. Review.
-
The nociceptin/orphanin FQ receptor (NOP) as a target for drug abuse medications.Curr Top Med Chem. 2011;11(9):1151-6. doi: 10.2174/156802611795371341. Curr Top Med Chem. 2011. PMID: 21050175 Free PMC article. Review.
-
Discovery and structure-activity relationships (SAR) of a novel class of 2-substituted N-piperidinyl indole-based nociceptin opioid receptor ligands.Bioorg Med Chem. 2023 Sep 7;92:117421. doi: 10.1016/j.bmc.2023.117421. Epub 2023 Jul 21. Bioorg Med Chem. 2023. PMID: 37573822 Free PMC article.
Cited by
-
Nociceptin/orphanin FQ receptor ligands and translational challenges: focus on cebranopadol as an innovative analgesic.Br J Anaesth. 2018 Nov;121(5):1105-1114. doi: 10.1016/j.bja.2018.06.024. Epub 2018 Aug 22. Br J Anaesth. 2018. PMID: 30336855 Free PMC article.
-
Anti-Parkinsonian and anti-dyskinetic profiles of two novel potent and selective nociceptin/orphanin FQ receptor agonists.Br J Pharmacol. 2018 Mar;175(5):782-796. doi: 10.1111/bph.14123. Epub 2018 Jan 31. Br J Pharmacol. 2018. PMID: 29232769 Free PMC article.
-
A Crosstalk between the Cannabinoid Receptors and Nociceptin Receptors in Colitis-Clinical Implications.J Clin Med. 2022 Nov 10;11(22):6675. doi: 10.3390/jcm11226675. J Clin Med. 2022. PMID: 36431153 Free PMC article. Review.
-
A novel orvinol analog, BU08028, as a safe opioid analgesic without abuse liability in primates.Proc Natl Acad Sci U S A. 2016 Sep 13;113(37):E5511-8. doi: 10.1073/pnas.1605295113. Epub 2016 Aug 29. Proc Natl Acad Sci U S A. 2016. PMID: 27573832 Free PMC article.
-
Endometriosis and Opioid Receptors: Are Opioids a Possible/Promising Treatment for Endometriosis?Int J Mol Sci. 2023 Jan 13;24(2):1633. doi: 10.3390/ijms24021633. Int J Mol Sci. 2023. PMID: 36675147 Free PMC article. Review.
References
-
- Mollereau C, Parmentier M, Mailleux P, Butour JL, Moisand C, Chalon P, Caput D, Vassart G, Meunier JC. ORL1, a novel member of the opioid receptor family. Cloning, functional expression and localization. FEBS Lett. 1994;341:33–38. - PubMed
-
- Bunzow JR, Saez C, Mortrud M, Bouvier C, Williams JT, Low M, Grandy DK. Molecular cloning and tissue distribution of a putative member of the rat opioid receptor gene family that is not a mu, delta or kappa opioid receptor type. FEBS Lett. 1994;347:284–288. - PubMed
-
- Fukuda K, Kato S, Mori K, Nishi M, Takeshima H, Iwabe N, Miyata T, Houtani T, Sugimoto T. cDNA cloning and regional distribution of a novel member of the opioid receptor family. FEBS Lett. 1994;343:42–46. - PubMed
-
- Wang JB, Johnson PS, Imai Y, Persico AM, Ozenberger BA, Eppler CM, Uhl GR. cDNA cloning of an orphan opiate receptor gene family member and its splice variant. FEBS Lett. 1994;348:75–79. - PubMed
-
- Pan YX, Cheng J, Xu J, Rossi G, Jacobson E, Ryan-Moro J, Brooks AI, Dean GE, Standifer KM, Pasternak GW. Cloning and functional characterization through antisense mapping of a kappa 3-related opioid receptor. Mol. Pharmacol. 1995;47:1180–1188. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

