Recommendations from the INHAND Apoptosis/Necrosis Working Group
- PMID: 26879688
- PMCID: PMC4785073
- DOI: 10.1177/0192623315625859
Recommendations from the INHAND Apoptosis/Necrosis Working Group
Abstract
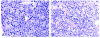
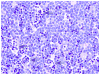
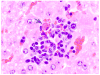
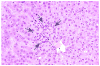
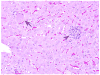
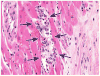
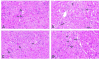
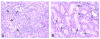
Historically, there has been confusion relating to the diagnostic nomenclature for individual cell death. Toxicologic pathologists have generally used the terms "single cell necrosis" and "apoptosis" interchangeably. Increased research on the mechanisms of cell death in recent years has led to the understanding that apoptosis and necrosis involve different cellular pathways and that these differences can have important implications when considering overall mechanisms of toxicity, and, for these reasons, the separate terms of apoptosis and necrosis should be used whenever differentiation is possible. However, it is also recognized that differentiation of the precise pathway of cell death may not be important, necessary, or possible in routine toxicity studies and so a more general term to indicate cell death is warranted in these situations. Morphological distinction between these two forms of cell death can sometimes be straightforward but can also be challenging. This article provides a brief discussion of the cellular mechanisms and morphological features of apoptosis and necrosis as well as guidance on when the pathologist should use these terms. It provides recommended nomenclature along with diagnostic criteria (in hematoxylin and eosin [H&E]-stained sections) for the most common forms of cell death (apoptosis and necrosis). This document is intended to serve as current guidance for the nomenclature of cell death for the International Harmonization of Nomenclature and Diagnostic Criteria Organ Working Groups and the toxicologic pathology community at large. The specific recommendations are:Use necrosis and apoptosis as separate diagnostic terms.Use modifiers to denote the distribution of necrosis (e.g., necrosis, single cell; necrosis, focal; necrosis, diffuse; etc.).Use the combined term apoptosis/single cell necrosis whenThere is no requirement or need to split the processes, orWhen the nature of cell death cannot be determined with certainty, orWhen both processes are present together. The diagnosis should be based primarily on the morphological features in H&E-stained sections. When needed, additional, special techniques to identify and characterize apoptosis can also be used.
Keywords: INHAND; apoptosis; cell death; guidance; necrosis; single cell necrosis.
© The Author(s) 2016.
Figures










Similar articles
-
International Harmonization of Nomenclature and Diagnostic Criteria (INHAND): Progress to Date and Future Plans.Toxicol Pathol. 2015 Jul;43(5):730-2. doi: 10.1177/0192623314560031. Epub 2014 Dec 21. Toxicol Pathol. 2015. PMID: 25530274
-
The nomenclature of cell death: recommendations of an ad hoc Committee of the Society of Toxicologic Pathologists.Toxicol Pathol. 1999 Jul-Aug;27(4):484-90. doi: 10.1177/019262339902700419. Toxicol Pathol. 1999. PMID: 10485836
-
International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) for Lesions in the Minipig.Toxicol Pathol. 2016 Apr;44(3):480-1. doi: 10.1177/0192623315614119. Epub 2015 Nov 3. Toxicol Pathol. 2016. PMID: 26534938
-
Brief Synopsis: Review of Renal Tubule and Interstitial Anatomy and Physiology and Renal INHAND, SEND, and DIKI Nomenclature.Toxicol Pathol. 2018 Dec;46(8):920-924. doi: 10.1177/0192623318798758. Epub 2018 Sep 6. Toxicol Pathol. 2018. PMID: 30189790 Review.
-
International harmonization of toxicologic pathology nomenclature: an overview and review of basic principles.Toxicol Pathol. 2012 Jun;40(4 Suppl):7S-13S. doi: 10.1177/0192623312438738. Toxicol Pathol. 2012. PMID: 22637736 Review.
Cited by
-
Cold Atmospheric Plasma induces accumulation of lysosomes and caspase-independent cell death in U373MG glioblastoma multiforme cells.Sci Rep. 2019 Sep 9;9(1):12891. doi: 10.1038/s41598-019-49013-3. Sci Rep. 2019. PMID: 31501494 Free PMC article.
-
A Review of Current Standards and the Evolution of Histopathology Nomenclature for Laboratory Animals.ILAR J. 2018 Dec 1;59(1):29-39. doi: 10.1093/ilar/ily005. ILAR J. 2018. PMID: 30476141 Free PMC article.
-
Calcium-Dependent Calpain Activation-Mediated Mitochondrial Dysfunction and Oxidative Stress Are Required for Cytotoxicity of Epinecidin-1 in Human Synovial Sarcoma SW982 Cells.Int J Mol Sci. 2020 Mar 19;21(6):2109. doi: 10.3390/ijms21062109. Int J Mol Sci. 2020. PMID: 32204400 Free PMC article.
-
ZBP1 not RIPK1 mediates tumor necroptosis in breast cancer.Nat Commun. 2021 May 11;12(1):2666. doi: 10.1038/s41467-021-23004-3. Nat Commun. 2021. PMID: 33976222 Free PMC article.
-
Development of an oral reference dose for the perfluorinated compound GenX.J Appl Toxicol. 2019 Sep;39(9):1267-1282. doi: 10.1002/jat.3812. Epub 2019 Jun 18. J Appl Toxicol. 2019. PMID: 31215065 Free PMC article.
References
-
- Chimal-Monroy J, Abarca-Buis RF, Cuervo R, Diaz-Hernandez M, Bustamante M, Rios-Flores A, Romero-Suarez S, Farrera-Hernandez A. Molecular control of cell differentiation and programmed cell death during digit development. IUBMB Life. 2011;63:922–9. - PubMed
-
- Dekkers MP, Barde YA. Developmental biology. Programmed cell death in neuronal development. Science. 2013;340:39–41. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

