Palladium-catalysed transannular C-H functionalization of alicyclic amines
- PMID: 26886789
- PMCID: PMC5082708
- DOI: 10.1038/nature16957
Palladium-catalysed transannular C-H functionalization of alicyclic amines
Abstract
Discovering pharmaceutical candidates is a resource-intensive enterprise that frequently requires the parallel synthesis of hundreds or even thousands of molecules. C-H bonds are present in almost all pharmaceutical agents. Consequently, the development of selective, rapid and efficient methods for converting these bonds into new chemical entities has the potential to streamline pharmaceutical development. Saturated nitrogen-containing heterocycles (alicyclic amines) feature prominently in pharmaceuticals, such as treatments for depression (paroxetine, amitifadine), diabetes (gliclazide), leukaemia (alvocidib), schizophrenia (risperidone, belaperidone), malaria (mefloquine) and nicotine addiction (cytisine, varenicline). However, existing methods for the C-H functionalization of saturated nitrogen heterocycles, particularly at sites remote to nitrogen, remain extremely limited. Here we report a transannular approach to selectively manipulate the C-H bonds of alicyclic amines at sites remote to nitrogen. Our reaction uses the boat conformation of the substrates to achieve palladium-catalysed amine-directed conversion of C-H bonds to C-C bonds on various alicyclic amine scaffolds. We demonstrate this approach by synthesizing new derivatives of several bioactive molecules, including varenicline.
Figures

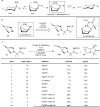


References
-
- Wencel-Delord J, Glorius F. C–H bond activation enables the rapid construction and late stage diversification of functional molecules. Nature Chem. 2013;5:369–375. - PubMed
-
- McMurray L, O'Hara F, Gaunt MJ. Recent developments in natural product synthesis using metal-catalysed C–H bond functionalization. Chem. Sov. Rev. 2011;40:1885–1898. - PubMed
-
- Yamaguchi J, Yamaguchi AD, Itami K. C-H bond functionalization: emerging synthetic tools for natural products and pharmaceuticals. Angew. Chem. Int. Ed. 2012;51:8960–9009. - PubMed
-
- Godula K, Sames D. C-H bond functionalization in complex organic synthesis. Science. 2006;312:67–72. - PubMed
-
- Taylor RD, MacCoss M, Lawson ADG. Rings in drugs. J. Med. Chem. 2014;57:5845–5859. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

