Impact of Aldosterone Antagonists on Sudden Cardiac Death Prevention in Heart Failure and Post-Myocardial Infarction Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
- PMID: 26891235
- PMCID: PMC4758660
- DOI: 10.1371/journal.pone.0145958
Impact of Aldosterone Antagonists on Sudden Cardiac Death Prevention in Heart Failure and Post-Myocardial Infarction Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
Background and objectives: Sudden cardiac death (SCD) is a severe burden of modern medicine. Aldosterone antagonist is publicized as effective in reducing mortality in patients with heart failure (HF) or post myocardial infarction (MI). Our study aimed to assess the efficacy of AAs on mortality including SCD, hospitalization admission and several common adverse effects.
Methods: We searched Embase, PubMed, Web of Science, Cochrane library and clinicaltrial.gov for randomized controlled trials (RCTs) assigning AAs in patients with HF or post MI through May 2015. The comparator included standard medication or placebo, or both. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed. Event rates were compared using a random effects model. Prospective RCTs of AAs with durations of at least 8 weeks were selected if they included at least one of the following outcomes: SCD, all-cause/cardiovascular mortality, all-cause/cardiovascular hospitalization and common side effects (hyperkalemia, renal function degradation and gynecomastia).
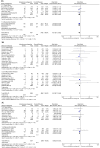
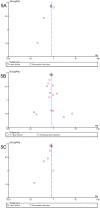
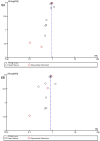
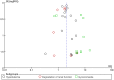
Results: Data from 19,333 patients enrolled in 25 trials were included. In patients with HF, this treatment significantly reduced the risk of SCD by 19% (RR 0.81; 95% CI, 0.67-0.98; p = 0.03); all-cause mortality by 19% (RR 0.81; 95% CI, 0.74-0.88, p<0.00001) and cardiovascular death by 21% (RR 0.79; 95% CI, 0.70-0.89, p<0.00001). In patients with post-MI, the matching reduced risks were 20% (RR 0.80; 95% CI, 0.66-0.98; p = 0.03), 15% (RR 0.85; 95% CI, 0.76-0.95, p = 0.003) and 17% (RR 0.83; 95% CI, 0.74-0.94, p = 0.003), respectively. Concerning both subgroups, the relative risks respectively decreased by 19% (RR 0.81; 95% CI, 0.71-0.92; p = 0.002) for SCD, 18% (RR 0.82; 95% CI, 0.77-0.88, p < 0.0001) for all-cause mortality and 20% (RR 0.80; 95% CI, 0.74-0.87, p < 0.0001) for cardiovascular mortality in patients treated with AAs. As well, hospitalizations were significantly reduced, while common adverse effects were significantly increased.
Conclusion: Aldosterone antagonists appear to be effective in reducing SCD and other mortality events, compared with placebo or standard medication in patients with HF and/or after a MI.
Conflict of interest statement
Figures



References
-
- Englestein E, Douglas Z. Sudden Cardiac Death, In: Alexander RWS, Schlant RC, Fuster V, The Heart, Arteries and Veins. (New York, NY: McGraw-Hill; ); 1998.
-
- Myerburg R, Castellanos A. Cardiac Arrest and Sudden Death, In: Braunwald E, Ed. Heart Disease: A Textbook of Cardiovascular Medicine., 9th edn Philadelphia: WB Saunders; 1997.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

