Influence of Molecular Structure on O2-Binding Properties and Blood Circulation of Hemoglobin‒Albumin Clusters
- PMID: 26895315
- PMCID: PMC4760709
- DOI: 10.1371/journal.pone.0149526
Influence of Molecular Structure on O2-Binding Properties and Blood Circulation of Hemoglobin‒Albumin Clusters
Abstract
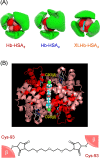
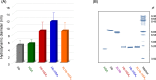
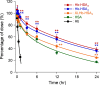
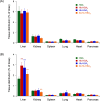
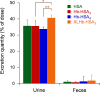
A hemoglobin wrapped covalently by three human serum albumins, a Hb-HSA3 cluster, is an artificial O2-carrier with the potential to function as a red blood cell substitute. This paper describes the synthesis and O2-binding properties of new hemoglobin‒albumin clusters (i) bearing four HSA units at the periphery (Hb-HSA4, large-size variant) and (ii) containing an intramolecularly crosslinked Hb in the center (XLHb-HSA3, high O2-affinity variant). Dynamic light scattering measurements revealed that the Hb-HSA4 diameter is greater than that of either Hb-HSA3 or XLHb-HSA3. The XLHb-HSA3 showed moderately high O2-affinity compared to the others because of the chemical linkage between the Cys-93(β) residues in Hb. Furthermore, the blood circulation behavior of 125I-labeled clusters was investigated by assay of blood retention and tissue distribution after intravenous administration into anesthetized rats. The XLHb-HSA3 was metabolized faster than Hb-HSA3 and Hb-HSA4. Results suggest that the molecular structure of the protein cluster is a factor that can influence in vivo circulation behavior.
Conflict of interest statement
Figures
Similar articles
-
Hemoglobin-albumin clusters as an artificial O2 carrier: Physicochemical properties and resuscitation from hemorrhagic shock in rats.J Biomed Mater Res B Appl Biomater. 2022 Aug;110(8):1827-1838. doi: 10.1002/jbm.b.35040. Epub 2022 Feb 22. J Biomed Mater Res B Appl Biomater. 2022. PMID: 35191606
-
Synthesis and physicochemical characterization of a series of hemoglobin-based oxygen carriers: objective comparison between cellular and acellular types.Bioconjug Chem. 2000 Jan-Feb;11(1):56-64. doi: 10.1021/bc9900789. Bioconjug Chem. 2000. PMID: 10639086
-
Hemoglobin-albumin cluster incorporating a Pt nanoparticle: artificial O2 carrier with antioxidant activities.PLoS One. 2014 Oct 13;9(10):e110541. doi: 10.1371/journal.pone.0110541. eCollection 2014. PLoS One. 2014. PMID: 25310133 Free PMC article.
-
Artificial oxygen carriers, hemoglobin vesicles and albumin-hemes, based on bioconjugate chemistry.Bioconjug Chem. 2009 Aug 19;20(8):1419-40. doi: 10.1021/bc800431d. Epub 2009 Feb 10. Bioconjug Chem. 2009. PMID: 19206516 Review.
-
Recent and prominent examples of nano- and microarchitectures as hemoglobin-based oxygen carriers.Adv Colloid Interface Sci. 2018 Oct;260:65-84. doi: 10.1016/j.cis.2018.08.006. Epub 2018 Aug 24. Adv Colloid Interface Sci. 2018. PMID: 30177214 Review.
Cited by
-
Pharmaceutical Integrity of Lyophilized Methemoglobin-Albumin Clusters after Reconstitution.ACS Omega. 2023 Jun 15;8(25):22589-22595. doi: 10.1021/acsomega.3c01054. eCollection 2023 Jun 27. ACS Omega. 2023. PMID: 37396217 Free PMC article.
-
Comparison of the Pharmacokinetic Properties of Hemoglobin-Based Oxygen Carriers.J Funct Biomater. 2017 Mar 18;8(1):11. doi: 10.3390/jfb8010011. J Funct Biomater. 2017. PMID: 28335469 Free PMC article. Review.
-
Artificial Blood for Dogs.Sci Rep. 2016 Nov 10;6:36782. doi: 10.1038/srep36782. Sci Rep. 2016. PMID: 27830776 Free PMC article.
References
-
- Bureau of Social Welfare and Public Health. Results on Blood Transfusion Situation 2013. Available: http://www.fukushihoken.metro.tokyo.jp/iryo/k_isyoku/yuketsutyousakekka.....
-
- Ministry of Health, Labor and Welfare, Japan. Proceedings of Blood Donation Promotion Committee, Pharmaceutical Affairs and Food Sanitation Council on 2 December, 2014. 2014. Available: http://www.mhlw.go.jp/file/05-Shingikai-11121000-Iyakushokuhinkyoku-Soum....
-
- Gould SA, Mass GS. Clinical development of human polymerized hemoglobin as a blood substitute. World J Surg. 1996;20: 1200–7. - PubMed
-
- Buehler PW, Boykins RA, Jia Y, Norris S, Freedberg DI, Alayash AI. Structural and functional characterization of glutaraldehyde-polymerized bovine hemoglobin and its isolated fractions. Anal Chem. 2005;77: 3466–78. - PubMed
-
- Pearce LB, Gawryl MS, Rentko VT, Moon-Massat PF, Rausch CW. HBOC-201 (hemoglobin glutamer-250) (bovine), Hemopure®): clinical studies In: Winslow RM, editor. Blood Substitutes. London: Elsevier; 2006. pp. 437–50.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

