Rosiglitazone Promotes Bone Marrow Adipogenesis to Impair Myelopoiesis under Stress
- PMID: 26895498
- PMCID: PMC4760757
- DOI: 10.1371/journal.pone.0149543
Rosiglitazone Promotes Bone Marrow Adipogenesis to Impair Myelopoiesis under Stress
Abstract
Objective: The therapeutic use of thiazolidinediones (TZDs) causes unwanted hematological side effects, although the underlying mechanisms of these effects are poorly understood. This study tests the hypothesis that rosiglitazone impairs the maintenance and differentiation of hematopoietic stem/progenitor cells, which ultimately leads to hematological abnormalities.
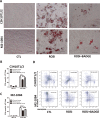
Methods: Mice were fed a rosiglitazone-supplemented diet or a normal diet for 6 weeks. To induce hematopoietic stress, all mice were injected once with 250 mg/kg 5-fluorouracil (5-Fu) intraperitoneally. Next, hematopoietic recovery, hematopoietic stem/progenitor cells (HSPCs) subsets, and myeloid differentiation after 5-Fu treatment were evaluated. The adipogenesis induced by rosiglitazone was assessed by histopathology and oil red O staining. The effect of adipocytes on HSPCs was studied with an in vitro co-culture system.
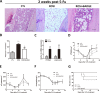
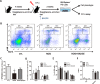
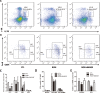
Results: Rosiglitazone significantly enhanced bone marrow adipogenesis and delayed hematopoietic recovery after 5-Fu treatment. Moreover, rosiglitazone inhibited proliferation of a granulocyte/monocyte progenitor (GMP) cell population and granulocyte/macrophage colony-stimulating factor (GM-CSF) colonies, although the proliferation and mobilization of Lin-c-kit+Sca-1+ cells (LSK) was maintained following hematopoietic stress. These effects could be partially reversed by the selective PPARγ antagonist BADGE. Finally, we demonstrated in a co-culture system that differentiated adipocytes actively suppressed the myeloid differentiation of HSPCs.
Conclusion: Taken together, our results demonstrate that rosiglitazone inhibits myeloid differentiation of HSPCs after stress partially by inducing bone marrow adipogenesis. Targeting the bone marrow microenvironment might be one mechanism by which rosiglitazone impairs stress-induced hematopoiesis.
Conflict of interest statement
Figures

Similar articles
-
Hematopoietic recovery following chemotherapy is improved by BADGE-induced inhibition of adipogenesis.Int J Hematol. 2013 Jan;97(1):58-72. doi: 10.1007/s12185-012-1233-4. Epub 2012 Dec 25. Int J Hematol. 2013. PMID: 23264188
-
Accelerated recovery of 5-fluorouracil-damaged bone marrow after rosiglitazone treatment.Eur J Pharmacol. 2005 Oct 17;522(1-3):122-9. doi: 10.1016/j.ejphar.2005.08.053. Epub 2005 Oct 6. Eur J Pharmacol. 2005. PMID: 16213483
-
Delineating the effects of 5-fluorouracil and follicle-stimulating hormone on mouse bone marrow stem/progenitor cells.Stem Cell Res Ther. 2016 Apr 19;7(1):59. doi: 10.1186/s13287-016-0311-6. Stem Cell Res Ther. 2016. PMID: 27095238 Free PMC article.
-
Regulation of myelopoiesis by proinflammatory cytokines in infectious diseases.Cell Mol Life Sci. 2018 Apr;75(8):1363-1376. doi: 10.1007/s00018-017-2724-5. Epub 2017 Dec 7. Cell Mol Life Sci. 2018. PMID: 29218601 Free PMC article. Review.
-
Roles of Hematopoietic Stem and Progenitor Cells in Ischemic Cardiovascular Disease.Curr Stem Cell Res Ther. 2021;16(5):589-598. doi: 10.2174/1574888X15666200130091858. Curr Stem Cell Res Ther. 2021. PMID: 32000654 Review.
Cited by
-
Mesenchymal stromal cells, metabolism, and mitochondrial transfer in bone marrow normal and malignant hematopoiesis.Front Cell Dev Biol. 2023 Dec 18;11:1325291. doi: 10.3389/fcell.2023.1325291. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 38169927 Free PMC article. Review.
-
Function and Regulation of Bone Marrow Adipose Tissue in Health and Disease: State of the Field and Clinical Considerations.Compr Physiol. 2024 Jun 27;14(3):5521-5579. doi: 10.1002/cphy.c230016. Compr Physiol. 2024. PMID: 39109972 Free PMC article. Review.
-
Pyrintegrin Induces Soft Tissue Formation by Transplanted or Endogenous Cells.Sci Rep. 2017 Jan 27;7:36402. doi: 10.1038/srep36402. Sci Rep. 2017. PMID: 28128224 Free PMC article.
-
Bone circuitry and interorgan skeletal crosstalk.Elife. 2023 Jan 19;12:e83142. doi: 10.7554/eLife.83142. Elife. 2023. PMID: 36656634 Free PMC article.
-
Label-free quantitative proteomics identifies transforming growth factor β1 (TGF-β1) as an inhibitor of adipogenic transformation in OP9-DL1 cells and primary thymic stromal cells.Cell Biosci. 2019 Jun 14;9:48. doi: 10.1186/s13578-019-0311-1. eCollection 2019. Cell Biosci. 2019. PMID: 31249661 Free PMC article.
References
-
- Clevidence DE, Juckett MB, Lucarelli MJ. Marrow suppression with myelodysplastic features, hypoerythropoetinemia, and lipotrophicproptosis due to rosiglitazone. WMJ. 2009;108: 462–465. - PubMed
-
- Berria R, Gastaldelli A, Lucidi S, Belfort R, Defilippis E, Easton C, et al. Reduction in hematocrit level after pioglitazone treatment is correlated with decreased plasma free testosterone level, not hemodilution, in women with polycystic ovary syndrome. Clin Pharmacol Ther. 2006;80: 105–114. 10.1016/j.clpt.2006.03.014 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

