Synchrotron Infrared and Deep UV Fluorescent Microspectroscopy Study of PB1-F2 β-Aggregated Structures in Influenza A Virus-infected Cells
- PMID: 26896002
- PMCID: PMC4861475
- DOI: 10.1074/jbc.M115.710533
Synchrotron Infrared and Deep UV Fluorescent Microspectroscopy Study of PB1-F2 β-Aggregated Structures in Influenza A Virus-infected Cells
Abstract
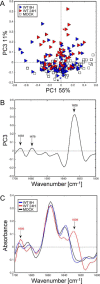
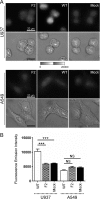
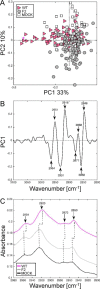
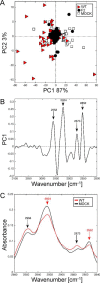
PB1-F2 is a virulence factor of influenza A virus (IAV) whose functions remain misunderstood. The different roles of PB1-F2 may be linked to its structural polymorphism and to its propensity to assemble into oligomers and amyloid fibers in the vicinity of the membrane of IAV-infected cells. Here, we monitored the impact of PB1-F2 on the biochemical composition and protein structures of human epithelial pulmonary cells (A549) and monocytic cells (U937) upon IAV infection using synchrotron Fourier-transform infrared (FTIR) and deep UV (DUV) microscopies at the single-cell level. Cells were infected with a wild-type IAV and its PB1-F2 knock-out mutant for analyses at different times post-infection. IR spectra were recorded in each condition and processed to evaluate the change in the component band of the spectra corresponding to the amide I (secondary structure) and the CH stretching region (membrane). The IR spectra analysis revealed that expression of PB1-F2 in U937 cells, but not in A549 cells, results in the presence of a specific β-aggregate signature. Furthermore, the lipid membrane composition of U937 cells expressing PB1-F2 was also altered in a cell type-dependent manner. Using DUV microscopy and taking advantage of the high content of tryptophan residues in the sequence of PB1-F2 (5/90 aa), we showed that the increase of the autofluorescent signal recorded in monocytic cells could be correlated with the IR detection of β-aggregates. Altogether, our results constitute an important step forward in the understanding of the cell type-dependent function of PB1-F2.
Keywords: Fourier transform IR (FTIR); amyloid; fluorescence imaging; influenza virus; membrane; microscopy; oligomerization; synchrotron radiation experiment.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures





References
-
- World Health Organization (2014) Global Influenza Surveillance and Response System (GISRS), http://www.who.int/influenza/gisrs_laboratory/en
-
- Kawaoka Y. (ed) (2006) Influenza Virology: Current Topics, Caister Academic Press, Wymondham
-
- Zell R., Krumbholz A., Eitner A., Krieg R., Halbhuber K. J., and Wutzler P. (2007) Prevalence of PB1-F2 of influenza A viruses. J. Gen. Virol. 88, 536–546 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

