Selective inhibition of tumor cell associated Vacuolar-ATPase 'a2' isoform overcomes cisplatin resistance in ovarian cancer cells
- PMID: 26899534
- PMCID: PMC5423172
- DOI: 10.1016/j.molonc.2016.01.003
Selective inhibition of tumor cell associated Vacuolar-ATPase 'a2' isoform overcomes cisplatin resistance in ovarian cancer cells
Abstract
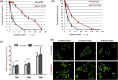
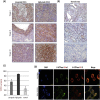
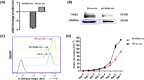
Development of resistance to platinum compounds significantly hinders successful ovarian cancer (OVCA) treatment. In tumor cells, dysregulated pH gradient across cell membranes is a key physiological mechanism of metastasis/chemo-resistance. These pH alterations are mediated by aberrant activation of key multi-subunit proton pumps, Vacuolar-ATPases (V-ATPases). In tumor cells, its 'a2' isoform (V-ATPase-V0a2) is a component of functional plasma-membrane complex and promotes tumor invasion through tumor-acidification and immuno-modulation. Its involvement in chemo-resistance has not been studied. Here, we show that V-ATPase-V0a2 is over-expressed in acquired-cisplatin resistant OVCA cells (cis-A2780/cis-TOV112D). Of all the 'a' subunit isoforms, V-ATPase-V0a2 exhibited an elevated expression on plasma membrane of cisplatin-resistant cells compared to sensitive counterparts. Immuno-histochemistry revealed V-ATPase-V0a2 expression in both low grade (highly drug-resistant) and high grade (highly recurrent) human OVCA tissues indicating its role in a centralized mechanism of tumor resistance. In cisplatin resistant cells, shRNA mediated inhibition of V-ATPase-V0a2 enhanced sensitivity towards both cisplatin and carboplatin. This improved cytotoxicity was mediated by enhanced cisplatin-DNA-adduct formation and suppressed DNA-repair pathway, leading to enhanced apoptosis. Suppression of V0a2 activity strongly reduced cytosolic pH in resistant tumor cells, which is known to enhance platinum-associated DNA-damage. As an indicator of reduced metastasis and chemo-resistance, in contrast to plasma membrane localization, a diffused cytoplasmic localization of acidic vacuoles was observed in V0a2-knockdown resistant cells. Interestingly, pre-treatment with monoclonal V0a2-inhibitory antibody enhanced cisplatin cytotoxicity in resistant cells. Taken together, our findings suggest that the isoform specific inhibition of V-ATPase-V0a2 could serve as a therapeutic strategy for chemo-resistant ovarian carcinoma and improve efficacy of platinum drugs.
Keywords: Cisplatin resistance; Ovarian cancer; Vacuolar ATPase; a2 isoform.
Copyright © 2016 Federation of European Biochemical Societies. Published by Elsevier B.V. All rights reserved.
Figures





Similar articles
-
In vivo anti-V-ATPase antibody treatment delays ovarian tumor growth by increasing antitumor immune responses.Mol Oncol. 2020 Oct;14(10):2436-2454. doi: 10.1002/1878-0261.12782. Epub 2020 Sep 10. Mol Oncol. 2020. PMID: 32797726 Free PMC article.
-
Targeting V-ATPase Isoform Restores Cisplatin Activity in Resistant Ovarian Cancer: Inhibition of Autophagy, Endosome Function, and ERK/MEK Pathway.J Oncol. 2019 Apr 1;2019:2343876. doi: 10.1155/2019/2343876. eCollection 2019. J Oncol. 2019. PMID: 31057611 Free PMC article.
-
Vacuolar ATPase 'a2' isoform exhibits distinct cell surface accumulation and modulates matrix metalloproteinase activity in ovarian cancer.Oncotarget. 2015 Feb 28;6(6):3797-810. doi: 10.18632/oncotarget.2902. Oncotarget. 2015. PMID: 25686833 Free PMC article.
-
Vacuolar ATPase as a potential therapeutic target and mediator of treatment resistance in cancer.Cancer Med. 2018 Aug;7(8):3800-3811. doi: 10.1002/cam4.1594. Epub 2018 Jun 21. Cancer Med. 2018. PMID: 29926527 Free PMC article. Review.
-
Chemoresistance in Ovarian Cancer: Prospects for New Drugs.Anticancer Agents Med Chem. 2021;21(6):668-678. doi: 10.2174/1871520620666200908104835. Anticancer Agents Med Chem. 2021. PMID: 32900355 Review.
Cited by
-
In vivo anti-V-ATPase antibody treatment delays ovarian tumor growth by increasing antitumor immune responses.Mol Oncol. 2020 Oct;14(10):2436-2454. doi: 10.1002/1878-0261.12782. Epub 2020 Sep 10. Mol Oncol. 2020. PMID: 32797726 Free PMC article.
-
The Golgi as a "Proton Sink" in Cancer.Front Cell Dev Biol. 2021 May 13;9:664295. doi: 10.3389/fcell.2021.664295. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34055797 Free PMC article.
-
Serum- and glucocorticoid- inducible kinase 2, SGK2, is a novel autophagy regulator and modulates platinum drugs response in cancer cells.Oncogene. 2020 Oct;39(40):6370-6386. doi: 10.1038/s41388-020-01433-6. Epub 2020 Aug 27. Oncogene. 2020. PMID: 32848212 Free PMC article.
-
V-ATPase in cancer progression: Two sides of the same coin.Oncotarget. 2018 Oct 16;9(81):35197-35198. doi: 10.18632/oncotarget.26218. eCollection 2018 Oct 16. Oncotarget. 2018. PMID: 30443284 Free PMC article. No abstract available.
-
Multifactorial Mechanisms of Tolerance to Ketoconazole in Candida albicans.Microbiol Spectr. 2021 Sep 3;9(1):e0032121. doi: 10.1128/Spectrum.00321-21. Epub 2021 Jun 23. Microbiol Spectr. 2021. PMID: 34160280 Free PMC article.
References
-
- Altomare, D.A. , 2004. AKT and mTOR phosphorylation is frequently detected in ovarian cancer and can be targeted to disrupt ovarian tumor cell growth. Oncogene. 23, 5853–5857. - PubMed
-
- American Cancer Society Statistics for 2014. [www.cancer.org].
-
- Beyenbach, K.W. , Wieczorek, H. , 2006. The V-type H+ ATPase: molecular structure and function, physiological roles and regulation. J. Exp. Biol. 209, 577–589. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

