Current state in the development of candidate therapeutic HPV vaccines
- PMID: 26901118
- PMCID: PMC4977850
- DOI: 10.1586/14760584.2016.1157477
Current state in the development of candidate therapeutic HPV vaccines
Abstract
The identification of human papillomavirus (HPV) as an etiological factor for HPV-associated malignancies creates the opportunity to control these cancers through vaccination. Currently, available preventive HPV vaccines have not yet demonstrated strong evidences for therapeutic effects against established HPV infections and lesions. Furthermore, HPV infections remain extremely common. Thus, there is urgent need for therapeutic vaccines to treat existing HPV infections and HPV-associated diseases. Therapeutic vaccines differ from preventive vaccines in that they are aimed at generating cell-mediated immunity rather than neutralizing antibodies. The HPV-encoded early proteins, especially oncoproteins E6 and E7, form ideal targets for therapeutic HPV vaccines since they are consistently expressed in HPV-associated malignancies and precancerous lesions, playing crucial roles in the generation and maintenance of HPV-associated disease. Our review will cover various therapeutic vaccines in development for the treatment of HPV-associated lesions and cancers. Furthermore, we review strategies to enhance vaccine efficacy and the latest clinical trials on therapeutic HPV vaccines.
Keywords: HPV; HPV E6; HPV E7; adjuvant; cervical cancer; clinical trials; combinatorial approach; human papillomavirus; immunotherapy; therapeutic vaccines.
Figures
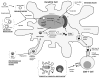
Similar articles
-
The current state of therapeutic and T cell-based vaccines against human papillomaviruses.Virus Res. 2017 Mar 2;231:148-165. doi: 10.1016/j.virusres.2016.12.002. Epub 2016 Dec 6. Virus Res. 2017. PMID: 27932207 Free PMC article. Review.
-
Perspectives for therapeutic HPV vaccine development.J Biomed Sci. 2016 Nov 4;23(1):75. doi: 10.1186/s12929-016-0293-9. J Biomed Sci. 2016. PMID: 27809842 Free PMC article. Review.
-
Advances in peptide-based human papillomavirus therapeutic vaccines.Curr Top Med Chem. 2012;12(14):1581-92. doi: 10.2174/156802612802652402. Curr Top Med Chem. 2012. PMID: 22827526 Review.
-
Therapeutic DNA Vaccines for Human Papillomavirus and Associated Diseases.Hum Gene Ther. 2018 Sep;29(9):971-996. doi: 10.1089/hum.2017.197. Epub 2018 Mar 16. Hum Gene Ther. 2018. PMID: 29316817 Free PMC article.
-
The Use of Both Therapeutic and Prophylactic Vaccines in the Therapy of Papillomavirus Disease.Front Immunol. 2020 Feb 18;11:188. doi: 10.3389/fimmu.2020.00188. eCollection 2020. Front Immunol. 2020. PMID: 32133000 Free PMC article. Review.
Cited by
-
Lipid Nanoparticles Outperform Electroporation in Delivering Therapeutic HPV DNA Vaccines.Vaccines (Basel). 2024 Jun 17;12(6):666. doi: 10.3390/vaccines12060666. Vaccines (Basel). 2024. PMID: 38932395 Free PMC article.
-
Smarter cures to combat COVID-19 and future pathogens: a review.Environ Chem Lett. 2021;19(4):2759-2771. doi: 10.1007/s10311-021-01224-9. Epub 2021 Apr 2. Environ Chem Lett. 2021. PMID: 33824633 Free PMC article. Review.
-
[Human papillomavirus and penile cancer : Thinking about measures for prevention].Urologe A. 2018 Apr;57(4):413-417. doi: 10.1007/s00120-018-0597-3. Urologe A. 2018. PMID: 29484460 Review. German.
-
Human Papillomavirus E6 and E7: The Cervical Cancer Hallmarks and Targets for Therapy.Front Microbiol. 2020 Jan 21;10:3116. doi: 10.3389/fmicb.2019.03116. eCollection 2019. Front Microbiol. 2020. PMID: 32038557 Free PMC article. Review.
-
Human Papillomavirus Genotype Detection in Oral Gargle Samples Among Men With Newly Diagnosed Oropharyngeal Squamous Cell Carcinoma.JAMA Otolaryngol Head Neck Surg. 2019 May 1;145(5):460-466. doi: 10.1001/jamaoto.2019.0119. JAMA Otolaryngol Head Neck Surg. 2019. PMID: 30920604 Free PMC article.
References
-
- Wakeham K, Kavanagh K. The burden of HPV-associated anogenital cancers. Current oncology reports. 2014;16(9):402. - PubMed
-
- Forman D, de Martel C, Lacey CJ, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012;30(Suppl 5):F12–23. - PubMed
-
- Mehanna H, Beech T, Nicholson T, et al. Prevalence of human papillomavirus in oropharyngeal and nonoropharyngeal head and neck cancer--systematic review and meta-analysis of trends by time and region. Head & neck. 2013;35(5):747–755. - PubMed
-
- Harper DM. Currently approved prophylactic HPV vaccines. Expert review of vaccines. 2009;8(12):1663–1679. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
