Tissue-Specific Signatures in the Transcriptional Response to Anaplasma phagocytophilum Infection of Ixodes scapularis and Ixodes ricinus Tick Cell Lines
- PMID: 26904518
- PMCID: PMC4748044
- DOI: 10.3389/fcimb.2016.00020
Tissue-Specific Signatures in the Transcriptional Response to Anaplasma phagocytophilum Infection of Ixodes scapularis and Ixodes ricinus Tick Cell Lines
Abstract
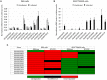
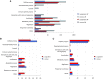
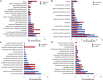
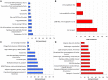
Anaplasma phagocytophilum are transmitted by Ixodes spp. ticks and have become one of the most common and relevant tick-borne pathogens due to their impact on human and animal health. Recent results have increased our understanding of the molecular interactions between Ixodes scapularis and A. phagocytophilum through the demonstration of tissue-specific molecular pathways that ensure pathogen infection, development and transmission by ticks. However, little is known about the Ixodes ricinus genes and proteins involved in the response to A. phagocytophilum infection. The tick species I. scapularis and I. ricinus are evolutionarily closely related and therefore similar responses are expected in A. phagocytophilum-infected cells. However, differences may exist between I. scapularis ISE6 and I. ricinus IRE/CTVM20 tick cells associated with tissue-specific signatures of these cell lines. To address this hypothesis, the transcriptional response to A. phagocytophilum infection was characterized by RNA sequencing and compared between I. scapularis ISE6 and I. ricinus IRE/CTVM20 tick cell lines. The transcriptional response to infection of I. scapularis ISE6 cells resembled that of tick hemocytes while the response in I. ricinus IRE/CTVM20 cells was more closely related to that reported previously in infected tick midguts. The inhibition of cell apoptosis by A. phagocytophilum appears to be a key adaptation mechanism to facilitate infection of both vertebrate and tick cells and was used to investigate further the tissue-specific response of tick cell lines to pathogen infection. The results supported a role for the intrinsic pathway in the inhibition of cell apoptosis by A. phagocytophilum infection of I. scapularis ISE6 cells. In contrast, the results in I. ricinus IRE/CTVM20 cells were similar to those obtained in tick midguts and suggested a role for the JAK/STAT pathway in the inhibition of apoptosis in tick cells infected with A. phagocytophilum. Nevertheless, tick cell lines were derived from embryonated eggs and may contain various cell populations with different morphology and behavior that could affect transcriptional response to infection. These results suggested tissue-specific signatures in I. scapularis ISE6 and I. ricinus IRE/CTVM20 tick cell line response to A. phagocytophilum infection that support their use as models for the study of tick-pathogen interactions.
Keywords: anaplasma; apoptosis; rickettsia; tick; tick cell line; transcriptomics.
Figures


References
-
- Alberdi P., Ayllón N., Cabezas-Cruz A., Bell-Sakyi L., Zweygarth E., Stuen S., et al. (2015). Infection of Ixodes spp. tick cells with different Anaplasma phagocytophilum isolates induces the inhibition of apoptotic cell death. Ticks Tick Borne Dis. 6, 758–767. 10.1016/j.ttbdis.2015.07.001 - DOI - PubMed
-
- Ayllón N., Naranjo V., Hajdušek O., Villar M., Galindo R. C., Kocan K. M., et al. (2015b). Nuclease Tudor-SN is involved in tick dsRNA-mediated RNA interference and feeding but not in defense against flaviviral or Anaplasma phagocytophilum rickettsial infection. PLoS ONE 10:e0133038. 10.1371/journal.pone.0133038 - DOI - PMC - PubMed
-
- Ayllón N., Villar V., Galindo R. C., Kocan K. M., Šíma R., López J. A., et al. (2015a). Systems biology of tissue-specific response to Anaplasma phagocytophilum reveals differentiated apoptosis in the tick vector Ixodes scapularis. PLoS Genet. 11:e1005120. 10.1371/journal.pgen.1005120 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

