Pharmacologic Comparison of Clinical Neutral Endopeptidase Inhibitors in a Rat Model of Acute Secretory Diarrhea
- PMID: 26907621
- PMCID: PMC4851326
- DOI: 10.1124/jpet.115.231167
Pharmacologic Comparison of Clinical Neutral Endopeptidase Inhibitors in a Rat Model of Acute Secretory Diarrhea
Abstract
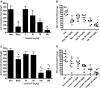
Racecadotril (acetorphan) is a neutral endopeptidase (NEP) inhibitor with known antidiarrheal activity in animals and humans; however, in humans, it suffers from shortcomings that might be improved with newer drugs in this class that have progressed to the clinic for nonenteric disease indications. To identify potentially superior NEP inhibitors with immediate clinical utility for diarrhea treatment, we compared their efficacy and pharmacologic properties in a rat intestinal hypersecretion model. Racecadotril and seven other clinical-stage inhibitors of NEP were obtained or synthesized. Enzyme potency and specificity were compared using purified peptidases. Compounds were orally administered to rats before administration of castor oil to induce diarrhea. Stool weight was recorded over 4 hours. To assess other pharmacologic properties, select compounds were orally administered to normal or castor oil-treated rats, blood and tissue samples collected at multiple time points, and active compound concentrations determined by mass spectroscopy. NEP enzyme activity was measured in tissue homogenates. Three previously untested clinical NEP inhibitors delayed diarrhea onset and reduced total stool output, with little or no effect on intestinal motility assessed by the charcoal meal test. Each was shown to be a potent, highly specific inhibitor of NEP. Each exhibited greater suppression of NEP activity in intestinal and nonintestinal tissues than did racecadotril and sustained this inhibition longer. These results suggest that newer clinical-stage NEP inhibitors originally developed for other indications may be directly repositioned for treatment of acute secretory diarrhea and offer advantages over racecadotril, such as less frequent dosing and potentially improved efficacy.
Copyright © 2016 The Author(s).
Figures
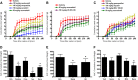
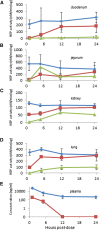
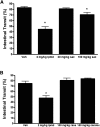
 ), 100 mg/kg racecadotril (
), 100 mg/kg racecadotril ( ), or 100 mg/kg sacubitril (
), or 100 mg/kg sacubitril ( ) was administered to normal healthy rats by oral gavage. At each time point, samples were collected from each indicated organ tissue for measurement of NEP enzyme activity. (E) At the same time points, the active metabolites of racecadotril (thiorphan,
) was administered to normal healthy rats by oral gavage. At each time point, samples were collected from each indicated organ tissue for measurement of NEP enzyme activity. (E) At the same time points, the active metabolites of racecadotril (thiorphan,  ) and sacubitril (LBQ657,
) and sacubitril (LBQ657,  ) were determined in plasma samples as described in Materials and Methods. Data are expressed as the mean ± S.E.M of values from four rats.
) were determined in plasma samples as described in Materials and Methods. Data are expressed as the mean ± S.E.M of values from four rats.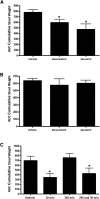
Similar articles
-
Drugs in Focus: The Use of Racecadotril in Paediatric Gastrointestinal Disease.J Pediatr Gastroenterol Nutr. 2020 Feb;70(2):162-164. doi: 10.1097/MPG.0000000000002574. J Pediatr Gastroenterol Nutr. 2020. PMID: 31978009
-
Racecadotril in the treatment of acute watery diarrhea in children.N Engl J Med. 2000 Aug 17;343(7):463-7. doi: 10.1056/NEJM200008173430703. N Engl J Med. 2000. PMID: 10944563 Clinical Trial.
-
Freeze dried extracts of Bidens biternata (Lour.) Merr. and Sheriff. show significant antidiarrheal activity in in-vivo models of diarrhea.J Ethnopharmacol. 2016 Dec 4;193:416-422. doi: 10.1016/j.jep.2016.09.041. Epub 2016 Sep 21. J Ethnopharmacol. 2016. PMID: 27664442
-
Racecadotril: a new approach to the treatment of diarrhoea.Int J Antimicrob Agents. 2000 Feb;14(1):75-9. doi: 10.1016/s0924-8579(99)00151-x. Int J Antimicrob Agents. 2000. PMID: 10717505 Review.
-
Racecadotril.Drugs. 2000 Apr;59(4):829-35; discussion 836-7. doi: 10.2165/00003495-200059040-00010. Drugs. 2000. PMID: 10804038 Review.
Cited by
-
Targeting Neprilysin (NEP) pathways: A potential new hope to defeat COVID-19 ghost.Biochem Pharmacol. 2020 Aug;178:114057. doi: 10.1016/j.bcp.2020.114057. Epub 2020 May 27. Biochem Pharmacol. 2020. PMID: 32470547 Free PMC article. Review.
-
Effects of Racecadotril on Weight Loss and Diarrhea Due to Human Rotavirus in Neonatal Gnotobiotic Pigs (Sus scrofa domesticus).Comp Med. 2017 Mar 1;67(2):157-164. Comp Med. 2017. PMID: 28381316 Free PMC article.
-
Molecular Basis for Omapatrilat and Sampatrilat Binding to Neprilysin-Implications for Dual Inhibitor Design with Angiotensin-Converting Enzyme.J Med Chem. 2020 May 28;63(10):5488-5500. doi: 10.1021/acs.jmedchem.0c00441. Epub 2020 May 8. J Med Chem. 2020. PMID: 32337993 Free PMC article.
-
The Active Ingredients Identification and Antidiarrheal Mechanism Analysis of Plantago asiatica L. Superfine Powder.Front Pharmacol. 2021 Jan 19;11:612478. doi: 10.3389/fphar.2020.612478. eCollection 2020. Front Pharmacol. 2021. PMID: 33542689 Free PMC article.
References
-
- Arnal JF, Castano C, Maupas E, Mugniot A, Darblade B, Gourdy P, Michel JB, Bayard F. (2001) Omapatrilat, a dual angiotensin-converting enzyme and neutral endopeptidase inhibitor, prevents fatty streak deposit in apolipoprotein E-deficient mice. Atherosclerosis 155:291–295. - PubMed
-
- Bergmann JF, Chaussade S, Couturier D, Baumer P, Schwartz JC, Lecomte JM. (1992) Effects of acetorphan, an antidiarrhoeal enkephalinase inhibitor, on oro-caecal and colonic transit times in healthy volunteers. Aliment Pharmacol Ther 6:305–313. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

