A critical period of vulnerability to adolescent stress: epigenetic mediators in mesocortical dopaminergic neurons
- PMID: 26908623
- PMCID: PMC4787906
- DOI: 10.1093/hmg/ddw019
A critical period of vulnerability to adolescent stress: epigenetic mediators in mesocortical dopaminergic neurons
Abstract
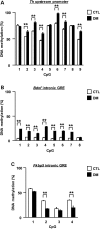
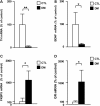
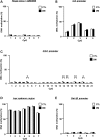
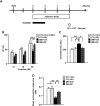
The molecular basis of vulnerability to stress during the adolescent period is largely unknown. To identify potential molecular mediators that may play a role in stress-induced behavioral deficits, we imposed social isolation on a genetically vulnerable mouse model. We report that 3-week (5-8 weeks of age) adolescent stress in combination with disrupted-in-schizophrenia 1 (Disc1) genetic risk elicits alterations in DNA methylation of a specific set of genes, tyrosine hydroxylase, brain-derived neurotrophic factor and FK506 binding protein 5. The epigenetic changes in the mesocortical dopaminergic neurons were prevented when animals were treated with a glucocorticoid receptor (GR) antagonist RU486 during social isolation, which implicates the role for glucocorticoid signaling in this pathological event. We define the critical period of GR intervention as the first 1-week period during the stress regimen, suggesting that this particular week in adolescence may be a specific period of maturation and function of mesocortical dopaminergic neurons and their sensitivity to glucocorticoids. Our study may also imply the clinical significance of early detection and prophylactic intervention against conditions associated with adolescent social stress in individuals with genetic risk.
© The Author 2016. Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures



References
-
- Blakemore S.J. (2008) The social brain in adolescence. Nat. Rev. Neurosci., 9, 267–277. - PubMed
-
- Caspi A., Roberts B.W., Shiner R.L. (2005) Personality development: stability and change. Ann. Rev. Psychol., 56, 453–484. - PubMed
-
- Gunnar M., Quevedo K. (2007) The neurobiology of stress and development. Ann. Rev. Psychol., 58, 145–173. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P50 MH094268/MH/NIMH NIH HHS/United States
- MH-069853/MH/NIMH NIH HHS/United States
- MH-092443/MH/NIMH NIH HHS/United States
- K99 MH094408/MH/NIMH NIH HHS/United States
- MH-084018/MH/NIMH NIH HHS/United States
- MH-085226/MH/NIMH NIH HHS/United States
- R21 DA040127/DA/NIDA NIH HHS/United States
- MH-094268/MH/NIMH NIH HHS/United States
- DA-040127/DA/NIDA NIH HHS/United States
- R01 MH092443/MH/NIMH NIH HHS/United States
- R00 MH093458/MH/NIMH NIH HHS/United States
- K99MH-094408/MH/NIMH NIH HHS/United States
- MH-088753/MH/NIMH NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

