Transfusion-Transmitted Dengue and Associated Clinical Symptoms During the 2012 Epidemic in Brazil
- PMID: 26908780
- PMCID: PMC4747611
- DOI: 10.1093/infdis/jiv326
Transfusion-Transmitted Dengue and Associated Clinical Symptoms During the 2012 Epidemic in Brazil
Abstract
Background: A linked donor-recipient study was conducted during epidemics in 2 cities in Brazil to investigate transfusion-transmitted (TT) dengue virus (DENV) by DENV RNA-positive donations.
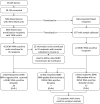
Methods: During February-June 2012, samples were collected from donors and recipients and retrospectively tested for DENV RNA by transcription-mediated amplification. Recipient chart review, using a case (DENV positive)-control (DENV negative and not known to be exposed) design, was conducted to assess symptoms.
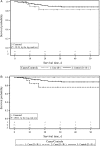
Results: Of 39 134 recruited blood donors, DENV-4 viremia was confirmed in 0.51% of donations from subjects in Rio de Janeiro and 0.80% of subjects in Recife. Overall, 42 DENV RNA-positive units were transfused into 35 recipients. Of these, 16 RNA-positive units transfused into 16 susceptible recipients were identified as informative: 5 cases were considered probable TT cases, 1 possible TT case, and 10 nontransmissions. The TT rate was 37.5% (95% confidence interval [CI], 15.2%-64.6%), significantly higher than the viremia rate of 0.93% (95% CI, .11%-3.34%) in nonexposed recipients (P < .0001). Chart review did not find significant differences between cases and controls in symptoms or mortality.
Conclusions: During a large epidemic of DENV-4 infection in Brazil, >0.5% of donations were RNA positive, and approximately one third of components resulted in TT. However, no significant clinical differences were evident between RNA-positive and RNA-negative recipients.
Keywords: NAT; clinical symptoms; dengue; transfusion-transmission.
© The Author 2015. Published by Oxford University Press for the Infectious Diseases Society of America. All rights reserved. For permissions, e-mail journals.permissions@oup.com.
Figures

Comment in
-
Dengue Virus and Blood Transfusion.J Infect Dis. 2016 Mar 1;213(5):689-90. doi: 10.1093/infdis/jiv322. Epub 2015 Jun 8. J Infect Dis. 2016. PMID: 26908779 No abstract available.
References
-
- Bianco C. Dengue and Chikungunya viruses in blood donations: risks to the blood supply? Transfusion 2008; 48:1279–81. - PubMed
-
- Chuang V, Wong TY, Leung YH et al. Review of dengue fever cases in Hong Kong during 1998 to 2005. Hong Kong Med J 2008; 14:170–7. - PubMed
-
- Holmes EC, Twiddy SS. The origin, emergence and evolutionary genetics of dengue virus. Infect Genet Evol 2003; 3:19–28. - PubMed
-
- Mackenzie JS, Gubler DJ, Petersen LR. Emerging flaviviruses: the spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nat Med 2004; 10:S98–109. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

