The role of the bone microenvironment in skeletal metastasis
- PMID: 26909265
- PMCID: PMC4723345
- DOI: 10.1016/j.jbo.2012.11.002
The role of the bone microenvironment in skeletal metastasis
Abstract
The bone microenvironment provides a fertile soil for cancer cells. It is therefore not surprising that the skeleton is a frequent site of cancer metastasis. It is believed that reciprocal interactions between tumour and bone cells, known as the "vicious cycle of bone metastasis" support the establishment and orchestrate the expansion of malignant cancers in bone. While the full range of molecular mechanisms of cancer metastasis to bone remain to be elucidated, recent research has deepened our understanding of the cell-mediated processes that may be involved in cancer cell survival and growth in bone. This review aims to address the importance of the bone microenvironment in skeletal cancer metastasis and discusses potential therapeutic implications of novel insights.
Keywords: Bone metastasis; Bone microenvironment; Bone remodelling; Cancer.
Figures

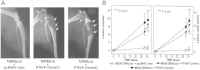

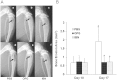
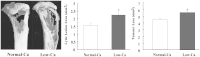
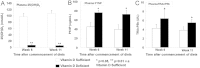

References
-
- Roodman GD. Mechanisms of bone metastasis. New England Journal of Medicine. 2004;350:1655–1664. - PubMed
-
- Coleman RE. Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer Treatment Reviews. 2001;27:165–176. - PubMed
-
- Fukutomi M, Yokota M, Chuman H, Harada H, Zaitsu Y, Funakoshi A. Increased incidence of bone metastases in hepatocellular carcinoma. European Journal of Gastroenterology and Hepatology. 2001;13:1083–1088. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

