NH125 kills methicillin-resistant Staphylococcus aureus persisters by lipid bilayer disruption
- PMID: 26910612
- PMCID: PMC4976882
- DOI: 10.4155/fmc.15.189
NH125 kills methicillin-resistant Staphylococcus aureus persisters by lipid bilayer disruption
Abstract
Background: NH125, a known WalK inhibitor kills MRSA persisters. However, its precise mode of action is still unknown.
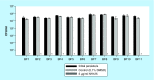
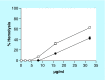
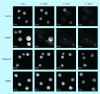
Methods & results: The mode of action of NH125 was investigated by comparing its spectrum of antimicrobial activity and its effects on membrane permeability and giant unilamellar vesicles (GUVs) with walrycin B, a WalR inhibitor and benzyldimethylhexadecylammonium chloride (16-BAC), a cationic surfactant. NH125 killed persister cells of a variety of Staphylococcus aureus strains. Similar to 16-BAC, NH125 killed MRSA persisters by inducing rapid membrane permeabilization and caused the rupture of GUVs, whereas walrycin B did not kill MRSA persisters or induce membrane permeabilization and did not affect GUVs.
Conclusion: NH125 kills MRSA persisters by interacting with and disrupting membranes in a detergent-like manner.
Keywords: MRSA; NH125; antibiotics; giant unilamellar vesicle; two-component system.
Conflict of interest statement
Financial & competing interests disclosure This study was supported by NIH grant P01 AI083214 to EE Mylonakis and FM Ausubel. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. No writing assistance was utilized in the production of this manuscript.
Figures


Similar articles
-
Antimicrobial peptide-inspired NH125 analogues: bacterial and fungal biofilm-eradicating agents and rapid killers of MRSA persisters.Org Biomol Chem. 2017 Jul 5;15(26):5503-5512. doi: 10.1039/c7ob01028a. Org Biomol Chem. 2017. PMID: 28534905
-
Identification of an Antimicrobial Agent Effective against Methicillin-Resistant Staphylococcus aureus Persisters Using a Fluorescence-Based Screening Strategy.PLoS One. 2015 Jun 3;10(6):e0127640. doi: 10.1371/journal.pone.0127640. eCollection 2015. PLoS One. 2015. PMID: 26039584 Free PMC article.
-
Identification of N-Arylated NH125 Analogues as Rapid Eradicating Agents against MRSA Persister Cells and Potent Biofilm Killers of Gram-Positive Pathogens.Chembiochem. 2017 Feb 16;18(4):352-357. doi: 10.1002/cbic.201600622. Epub 2017 Jan 16. Chembiochem. 2017. PMID: 27925693
-
Strategies against methicillin-resistant Staphylococcus aureus persisters.Future Med Chem. 2018 Apr 1;10(7):779-794. doi: 10.4155/fmc-2017-0199. Epub 2018 Mar 23. Future Med Chem. 2018. PMID: 29569952 Free PMC article. Review.
-
[Design, Synthesis and Structure-activity Relationship Study of a Series of Bis(bibenzyl)-type Natural Products, Riccardin C Derivatives, as Candidate Anti-MRSA Agents].Yakugaku Zasshi. 2018;138(12):1537-1547. doi: 10.1248/yakushi.18-00135. Yakugaku Zasshi. 2018. PMID: 30504670 Review. Japanese.
Cited by
-
Influence of subinhibitory concentrations of NH125 on biofilm formation & virulence factors of Staphylococcus aureus.Future Med Chem. 2018 Jun 1;10(11):1319-1331. doi: 10.4155/fmc-2017-0286. Epub 2018 May 30. Future Med Chem. 2018. PMID: 29846088 Free PMC article.
-
Bacterial persisters: molecular mechanisms and therapeutic development.Signal Transduct Target Ther. 2024 Jul 17;9(1):174. doi: 10.1038/s41392-024-01866-5. Signal Transduct Target Ther. 2024. PMID: 39013893 Free PMC article. Review.
-
Membrane depolarization kills dormant Bacillus subtilis cells by generating a lethal dose of ROS.Nat Commun. 2024 Aug 11;15(1):6877. doi: 10.1038/s41467-024-51347-0. Nat Commun. 2024. PMID: 39128925 Free PMC article.
-
In Vitro and In Vivo Bactericidal and Antibiofilm Efficacy of Alpha Mangostin Against Staphylococcus aureus Persister Cells.Front Cell Infect Microbiol. 2022 Jul 22;12:898794. doi: 10.3389/fcimb.2022.898794. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35937701 Free PMC article.
-
Essential Two-Component Systems Regulating Cell Envelope Functions: Opportunities for Novel Antibiotic Therapies.J Membr Biol. 2018 Feb;251(1):75-89. doi: 10.1007/s00232-017-9995-5. Epub 2017 Nov 2. J Membr Biol. 2018. PMID: 29098331 Review.
References
-
- Gorwitz RJ, Kruszon-Moran D, McAllister SK, et al. Changes in the prevalence of nasal colonization with Staphylococcus aureus in the United States, 2001–2004. J. Infect. Dis. 2008;197(9):1226–1234. - PubMed
-
- Wendlandt S, Schwarz S, Silley P. Methicillin-resistant Staphylococcus aureus: a food-borne pathogen? Annu. Rev. Food Sci. Technol. 2013;4(1):117–139. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
