Mutation Screening of Candidate Genes in Patients with Nonsyndromic Sagittal Craniosynostosis
- PMID: 26910679
- PMCID: PMC4770826
- DOI: 10.1097/01.prs.0000479978.75545.ee
Mutation Screening of Candidate Genes in Patients with Nonsyndromic Sagittal Craniosynostosis
Abstract
Background: Craniosynostosis is a condition that includes the premature fusion of one or multiple cranial sutures. Among various craniosynostosis forms, sagittal nonsyndromic craniosynostosis is the most prevalent. Although different gene mutations have been identified in some craniosynostosis syndromes, the cause of sagittal nonsyndromic craniosynostosis remains largely unknown.
Methods: To screen for candidate genes for sagittal nonsyndromic craniosynostosis, the authors sequenced DNA of 93 sagittal nonsyndromic craniosynostosis patients from a population-based study conducted in Iowa and New York states. FGFR1-3 mutational hotspots and the entire TWIST1, RAB23, and BMP2 coding regions were screened because of their known roles in human nonsyndromic or syndromic sagittal craniosynostosis, expression patterns, and/or animal model studies.
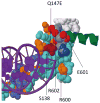
Results: The authors identified two rare variants in their cohort. A FGFR1 insertion c.730_731insG, which led to a premature stop codon, was predicted to abolish the entire immunoglobulin-like III domain, including the ligand-binding region. A c.439C>G variant was observed in TWIST1 at its highly conserved loop domain in another patient. The patient's mother harbored the same variant and was reported with jaw abnormalities. These two variants were not detected in 116 alleles from unaffected controls or seen in the several databases; however, TWIST1 variant was found in a low frequency of 0.000831 percent in Exome Aggregation Consortium database.
Conclusions: The low mutation detection rate indicates that these genes account for only a small proportion of sagittal nonsyndromic craniosynostosis patients. The authors' results add to the perception that sagittal nonsyndromic craniosynostosis is a complex developmental defect with considerable genetic heterogeneity.
Clinical question/level of evidence: Risk, II.
Figures
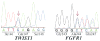

References
-
- Zeiger JS, Beaty TH, Hetmanski JB, et al. Genetic and environmental risk factors for sagittal craniosynostosis. J Craniofac Surg. 2002;13:602–606. - PubMed
-
- Boyadjiev SA International Craniosynostosis Consortium. Genetic analysis of non-syndromic craniosynostosis. Orthod Craniofac Res. 2007;10:129–137. - PubMed
-
- Gerety P, Basta MN, Fischer JP, Taylor JA, Bartlett SP. Operative management of non-syndromic sagittal craniosynostosis: A head-to-head meta-analysis of outcomes comparing three techniques. Plast Reconstr Surg. 2014;134:23–24.
-
- Lajeunie E, Le Merrer M, Bonaiti-Pellie C, Marchac D, Renier D. Genetic study of scaphocephaly. Am J Med Genet. 1996;62:282–285. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

