Glucose-regulated protein 94 deficiency induces squamous cell metaplasia and suppresses PTEN-null driven endometrial epithelial tumor development
- PMID: 26910913
- PMCID: PMC4924759
- DOI: 10.18632/oncotarget.7450
Glucose-regulated protein 94 deficiency induces squamous cell metaplasia and suppresses PTEN-null driven endometrial epithelial tumor development
Abstract
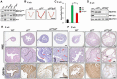
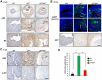
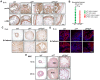
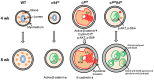
Endometrial carcinoma is the most prevalent gynecologic cancer in the United States. The tumor suppressor gene Pten (phosphatase and tensin homolog) is commonly mutated in the more common type 1 (endometrioid) subtype. The glucose-regulated protein 94 (GRP94) is emerging as a novel regulator for cancer development. Here we report that expression profiles from the Cancer Genome Atlas (TCGA) showed significantly increased Grp94 mRNA levels in endometrial tumor versus normal tissues, correlating with highly elevated GRP94 protein expression in patient samples and the requirement of GRP94 for maintaining viability of human endometrioid adenocarcinoma (EAC) cell lines. Through generation of uterus-specific knockout mouse models with deletion of Grp94 alone (c94f/f) or in combination with Pten (cPf/f94f/f), we discovered that c94f/f uteri induced squamous cell metaplasia (SCM) and reduced active nuclear β-catenin. The cPf/f94f/f uteri showed accelerated SCM and suppression of PTEN-null driven EAC, with reduced cellular proliferation, attenuated β-catenin signaling and decreased AKT/S6 activation in the SCM. In contrast to single PTEN knockout uteri (cPf/f), cPf/f94f/f uteri showed no decrease in E-cadherin level and no invasive lesion. Collectively, our study implies that GRP94 downregulation induces SCM in EAC and suppresses AKT/S6 signaling, providing a novel mechanism for suppressing EAC progression.
Keywords: PTEN; endometrial cancer; glucose-regulated protein 94 (GRP94); squamous cell metaplasia; β-catenin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Targeting the glucose-regulated protein-78 abrogates Pten-null driven AKT activation and endometrioid tumorigenesis.Oncogene. 2015 Oct;34(43):5418-26. doi: 10.1038/onc.2015.4. Epub 2015 Feb 16. Oncogene. 2015. PMID: 25684138 Free PMC article.
-
Alterations in Wnt-β-catenin and Pten signalling play distinct roles in endometrial cancer initiation and progression.J Pathol. 2013 May;230(1):48-58. doi: 10.1002/path.4160. Epub 2013 Mar 14. J Pathol. 2013. PMID: 23288720
-
[Expression of beta-catenin, Glut-1, PTEN proteins in uterine endometrioid adenocarcinoma and its precursor lesions].Zhonghua Bing Li Xue Za Zhi. 2009 Sep;38(9):594-9. Zhonghua Bing Li Xue Za Zhi. 2009. PMID: 20079187 Chinese.
-
Molecular pathology of endometrial carcinoma.Histopathology. 2013 Jan;62(1):111-23. doi: 10.1111/his.12053. Histopathology. 2013. PMID: 23240673 Review.
-
Immunophenotype of Atypical Polypoid Adenomyoma of the Uterus: Diagnostic Value and Insight on Pathogenesis.Appl Immunohistochem Mol Morphol. 2020 Sep;28(8):646-653. doi: 10.1097/PAI.0000000000000780. Appl Immunohistochem Mol Morphol. 2020. PMID: 31855579
Cited by
-
New role of endoplasmic reticulum chaperones in regulating metaplasia during tumorigenesis.Mol Cell Oncol. 2017 Jun 30;4(6):e1345350. doi: 10.1080/23723556.2017.1345350. eCollection 2017. Mol Cell Oncol. 2017. PMID: 29209644 Free PMC article.
-
Co-downregulation of GRP78 and GRP94 Induces Apoptosis and Inhibits Migration in Prostate Cancer Cells.Open Life Sci. 2019 Jul 22;14:384-391. doi: 10.1515/biol-2019-0043. eCollection 2019 Jan. Open Life Sci. 2019. PMID: 33817173 Free PMC article.
-
Co-existing TP53 and ARID1A mutations promote aggressive endometrial tumorigenesis.PLoS Genet. 2021 Dec 23;17(12):e1009986. doi: 10.1371/journal.pgen.1009986. eCollection 2021 Dec. PLoS Genet. 2021. PMID: 34941867 Free PMC article.
-
Impact of HSP90α, CEA, NSE, SCC, and CYFRA21-1 on Lung Cancer Patients.J Healthc Eng. 2021 Oct 20;2021:6929971. doi: 10.1155/2021/6929971. eCollection 2021. J Healthc Eng. 2021. Retraction in: J Healthc Eng. 2023 Oct 11;2023:9796148. doi: 10.1155/2023/9796148. PMID: 34721827 Free PMC article. Retracted.
-
Autophagy, molecular chaperones, and unfolded protein response as promoters of tumor recurrence.Cancer Metastasis Rev. 2023 Mar;42(1):217-254. doi: 10.1007/s10555-023-10085-3. Epub 2023 Feb 1. Cancer Metastasis Rev. 2023. PMID: 36723697 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

