A novel follicle-stimulating hormone receptor mutation causing primary ovarian failure: a fertility application of whole exome sequencing
- PMID: 26911863
- PMCID: PMC5007606
- DOI: 10.1093/humrep/dew025
A novel follicle-stimulating hormone receptor mutation causing primary ovarian failure: a fertility application of whole exome sequencing
Abstract
Study question: Can whole exome sequencing (WES) and in vitro validation studies be used to find the causative genetic etiology in a patient with primary ovarian failure and infertility?
Summary answer: A novel follicle-stimulating hormone receptor (FSHR) mutation was found by WES and shown, via in vitro flow cytometry studies, to affect membrane trafficking.
What is known already: WES may diagnose up to 25-35% of patients with suspected disorders of sex development (DSD). FSHR mutations are an extremely rare cause of 46, XX gonadal dysgenesis with primary amenorrhea due to hypergonadotropic ovarian failure.
Study design, size, duration: A WES study was followed by flow cytometry studies of mutant protein function.
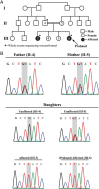
Participants/materials, setting, methods: The study subjects were two Turkish sisters with hypergonadotropic primary amenorrhea, their parents and two unaffected sisters. The affected siblings and both parents were sequenced (trio-WES). Transient transfection of HEK 293T cells was performed with a vector containing wild-type FSHR as well as the novel FSHR variant that was discovered by WES. Cellular localization of FSHR protein as well as FSH-stimulated cyclic AMP (cAMP) production was evaluated using flow cytometry.
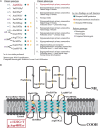
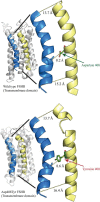
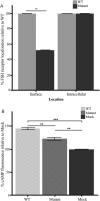
Main results and the role of chance: Both affected sisters were homozygous for a previously unreported missense mutation (c.1222G>T, p.Asp408Tyr) in the second transmembrane domain of FSHR. Modeling predicted disrupted secondary structure. Flow cytometry demonstrated an average of 48% reduction in cell-surface signal detection (P < 0.01). The mean fluorescent signal for cAMP (second messenger of FSHR), stimulated by FSH, was reduced by 50% in the mutant-transfected cells (P < 0.01).
Limitations, reasons for caution: This is an in vitro validation. All novel purported genetic variants can be clinically reported only as 'variants of uncertain significance' until more patients with a similar phenotype are discovered with the same variant.
Wider implications of the findings: We report the first WES-discovered FSHR mutation, validated by quantitative flow cytometry. WES is a valuable tool for diagnosis of rare genetic diseases, and flow cytometry allows for quantitative characterization of purported variants. WES-assisted diagnosis allows for treatments aimed at the underlying molecular etiology of disease. Future studies should focus on pharmacological and assisted reproductive treatments aimed at the disrupted FSHR, so that patients with FSH resistance can be treated by personalized medicine.
Study funding/competing interests: E.V. is partially funded by the DSD Translational Research Network (NICHD 1R01HD068138). M.S.B. is funded by the Neuroendocrinology, Sex Differences and Reproduction training grant (NICHD 5T32HD007228). The authors have no competing interests to disclose.
Keywords: follicle-stimulating hormone receptor; premature ovarian failure; primary amenorrhea; resistant ovary syndrome; whole exome sequencing.
© The Author 2016. Published by Oxford University Press on behalf of the European Society of Human Reproduction and Embryology. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures
Similar articles
-
Pathogenic variants in DLGAP5 cause female infertility characterized by oocyte maturation arrest and embryonic arrest.Hum Reprod. 2025 Jul 10:deaf139. doi: 10.1093/humrep/deaf139. Online ahead of print. Hum Reprod. 2025. PMID: 40639803
-
The interplay between mitochondrial DNA genotypes, female infertility, ovarian response, and mutagenesis in oocytes.Hum Reprod Open. 2024 Dec 30;2025(1):hoae074. doi: 10.1093/hropen/hoae074. eCollection 2025. Hum Reprod Open. 2024. PMID: 39830711 Free PMC article.
-
CCDC89 is required for optimal sperm motility and male fertility in mammals.Hum Reprod. 2025 Jul 1:deaf126. doi: 10.1093/humrep/deaf126. Online ahead of print. Hum Reprod. 2025. PMID: 40591933
-
Assessing the comparative effects of interventions in COPD: a tutorial on network meta-analysis for clinicians.Respir Res. 2024 Dec 21;25(1):438. doi: 10.1186/s12931-024-03056-x. Respir Res. 2024. PMID: 39709425 Free PMC article. Review.
-
Follicle stimulating hormone receptor in ovarian surface epithelium and epithelial ovarian cancer.Oncol Res. 2008;17(5):231-8. doi: 10.3727/096504008786111383. Oncol Res. 2008. PMID: 18980020
Cited by
-
The Effect of FSHR (G2039A) Polymorphism on Müllerian Duct Development and Hormonal Profile of Women with Primary Amenorrhea.J Reprod Infertil. 2023 Oct-Dec;24(4):240-247. doi: 10.18502/jri.v24i4.14151. J Reprod Infertil. 2023. PMID: 38164425 Free PMC article.
-
Identification and characterization of novel compound heterozygous variants in FSHR causing primary ovarian insufficiency with resistant ovary syndrome.Front Endocrinol (Lausanne). 2023 Jan 10;13:1013894. doi: 10.3389/fendo.2022.1013894. eCollection 2022. Front Endocrinol (Lausanne). 2023. PMID: 36704038 Free PMC article.
-
Genetics of Disorders of Sex Development: The DSD-TRN Experience.Endocrinol Metab Clin North Am. 2017 Jun;46(2):519-537. doi: 10.1016/j.ecl.2017.01.015. Epub 2017 Mar 28. Endocrinol Metab Clin North Am. 2017. PMID: 28476235 Free PMC article. Review.
-
The landscape of genetic variations in non-syndromic primary ovarian insufficiency in the MENA region: a systematic review.Front Endocrinol (Lausanne). 2024 Apr 26;14:1289333. doi: 10.3389/fendo.2023.1289333. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38737775 Free PMC article.
-
Misfolded G Protein-Coupled Receptors and Endocrine Disease. Molecular Mechanisms and Therapeutic Prospects.Int J Mol Sci. 2021 Nov 15;22(22):12329. doi: 10.3390/ijms222212329. Int J Mol Sci. 2021. PMID: 34830210 Free PMC article. Review.
References
-
- Aittomaki K, Lucena JL, Pakarinen P, Sistonen P, Tapanainen J, Gromoll J, Kaskikari R, Sankila EM, Lehvaslaiho H, Engel AR et al. Mutation in the follicle-stimulating hormone receptor gene causes hereditary hypergonadotropic ovarian failure. Cell 1995;82:959–968. - PubMed
-
- Aittomaki K, Herva R, Stenman UH, Juntunen K, Ylostalo P, Hovatta O, de la Chapelle A. Clinical features of primary ovarian failure caused by a point mutation in the follicle-stimulating hormone receptor gene. J Clin Endocrinol Metab 1996;81:3722–3726. - PubMed
-
- Allen LA, Achermann JC, Pakarinen P, Kotlar TJ, Huhtaniemi IT, Jameson JL, Cheetham TD, Ball SG. A novel loss of function mutation in exon 10 of the FSH receptor gene causing hypergonadotrophic hypogonadism: clinical and molecular characteristics. Hum Reprod 2003;18:251–256. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

