A Randomized, Controlled Trial of Oral Intestinal Sorbent AST-120 on Renal Function Deterioration in Patients with Advanced Renal Dysfunction
- PMID: 26912554
- PMCID: PMC4822676
- DOI: 10.2215/CJN.12011214
A Randomized, Controlled Trial of Oral Intestinal Sorbent AST-120 on Renal Function Deterioration in Patients with Advanced Renal Dysfunction
Abstract
Background and objectives: The notion that oral intestinal sorbent AST-120 slows renal disease progression has not been evaluated thoroughly. In this study, we investigated the long-term effect of AST-120 on renal disease progression (doubling of serum creatinine, eGFR decrease >50%, or initiation of RRT) in patients with advanced CKD.
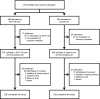
Design, setting, participants, & measurements: We prospectively recruited 579 patients (CKD stage 3 or 4) from 11 medical centers in Korea from March 4, 2009 to August 31, 2010 and randomized them into an AST-120 arm and a control arm. Patients in the AST-120 arm were given 6 g AST-120 in three divided doses per day, and those in the control arm received only standard conventional treatment (open-label design) for 36 months or until the occurrence of primary outcomes.
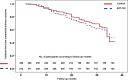
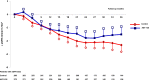
Results: Levels of serum and urine indoxyl sulfate and β2-microglobulin decreased throughout the study period in both treatment arms; however, there was not a significant difference in change in uremic toxins in the AST-120 and control arms. The two arms were not different in the occurrence of composite primary outcomes (100 events in 272 individuals in the AST-120 arm and 100 events in 266 individuals in the control arm; hazard ratio, 1.12; 95% confidence interval, 0.85 to 1.48; log-rank P=0.45). The decline in eGFR and change in proteinuria were similar in the two treatment arms over time (Prandomization-time=0.64 and Prandomization-time=0.16, respectively). There was no difference in mortality (nine deaths in the AST-120 arm and 11 deaths in the control arm; log-rank P=0.73) or unplanned hospitalizations (102 in the AST-120 arm and 109 in the control arm; log-rank P=0.76) in the two treatment arms. There was no significant difference of the health-related quality of life score between the two arms.
Conclusions: Long-term use of AST-120 added to standard treatment did not change renal disease progression, proteinuria, mortality, and health-related quality of life in patients with advanced renal dysfunction.
Keywords: Randomized trial; disease progression; glomerular filtration rate; hospitalization; humans; kidney; kidney function tests; proteinuria; renal insufficiency, chronic; uremic toxin.
Copyright © 2016 by the American Society of Nephrology.
Figures
References
-
- ESRD Registry Committee, Korean Society of Nephrology: Current status of renal replacement therapy in Korea—Insan Memorial Dialysis Registry 2012. Presented at the 33rd Annual Autumn Meeting of the Korean Society of Nephrology, Gwangju Metropolitan City, Republic of Korea, October 12, 2013
-
- Lee SW, Kim YC, Oh SW, Koo HS, Na KY, Chae DW, Kim S, Chin HJ: Trends in the prevalence of chronic kidney disease, other chronic diseases and health-related behaviors in an adult Korean population: Data from the Korean National Health and Nutrition Examination Survey (KNHANES). Nephrol Dial Transplant 26: 3975–3980, 2011 - PubMed
-
- Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, Ritz E, Atkins RC, Rohde R, Raz I, Collaborative Study Group : Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 345: 851–860, 2001 - PubMed
-
- Keane WF, Brenner BM, de Zeeuw D, Grunfeld JP, McGill J, Mitch WE, Ribeiro AB, Shahinfar S, Simpson RL, Snapinn SM, Toto R, RENAAL Study Investigators : The risk of developing end-stage renal disease in patients with type 2 diabetes and nephropathy: The RENAAL study. Kidney Int 63: 1499–1507, 2003 - PubMed
-
- Hou FF, Xie D, Zhang X, Chen PY, Zhang WR, Liang M, Guo ZJ, Jiang JP: Renoprotection of Optimal Antiproteinuric Doses (ROAD) Study: A randomized controlled study of benazepril and losartan in chronic renal insufficiency. J Am Soc Nephrol 18: 1889–1898, 2007 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

