γ-Crystallins of the chicken lens: remnants of an ancient vertebrate gene family in birds
- PMID: 26913478
- PMCID: PMC5576348
- DOI: 10.1111/febs.13689
γ-Crystallins of the chicken lens: remnants of an ancient vertebrate gene family in birds
Abstract
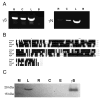
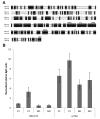
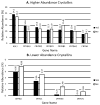
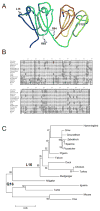
γ-Crystallins, abundant proteins of vertebrate lenses, were thought to be absent from birds. However, bird genomes contain well-conserved genes for γS- and γN-crystallins. Although expressed sequence tag analysis of chicken eye found no transcripts for these genes, RT-PCR detected spliced transcripts for both genes in chicken lens, with lower levels in cornea and retina/retinal pigment epithelium. The level of mRNA for γS in chicken lens was relatively very low even though the chicken crygs gene promoter had lens-preferred activity similar to that of mouse. Chicken γS was detected by a peptide antibody in lens, but not in other ocular tissues. Low levels of γS and γN proteins were detected in chicken lens by shotgun mass spectroscopy. Water-soluble and water-insoluble lens fractions were analyzed and 1934 proteins (< 1% false discovery rate) were detected, increasing the known chicken lens proteome 30-fold. Although chicken γS is well conserved in protein sequence, it has one notable difference in leucine 16, replacing a surface glutamine conserved in other γ-crystallins, possibly affecting solubility. However, L16 and engineered Q16 versions were both highly soluble and had indistinguishable circular dichroism, tryptophan fluorescence and heat stability (melting temperature Tm ~ 65 °C) profiles. L16 has been present in birds for over 100 million years and may have been adopted for a specific protein interaction in the bird lens. However, evolution has clearly reduced or eliminated expression of ancestral γ-crystallins in bird lenses. The conservation of genes for γS- and γN-crystallins suggests they may have been preserved for reasons unrelated to the bulk properties of the lens.
Keywords: crystallin; evolution; eye; promoter; protein folding; proteomics.
Published 2016. This article is a U.S. Government work and is in the public domain in the USA.
Figures

Similar articles
-
gammaN-crystallin and the evolution of the betagamma-crystallin superfamily in vertebrates.FEBS J. 2005 May;272(9):2276-91. doi: 10.1111/j.1742-4658.2005.04655.x. FEBS J. 2005. PMID: 15853812
-
Expressed sequence tag analysis of guinea pig (Cavia porcellus) eye tissues for NEIBank.Mol Vis. 2008;14:2413-27. Epub 2008 Dec 19. Mol Vis. 2008. PMID: 19104676 Free PMC article.
-
Expression and regulation of alpha-, beta-, and gamma-crystallins in mammalian lens epithelial cells.Invest Ophthalmol Vis Sci. 2004 Oct;45(10):3608-19. doi: 10.1167/iovs.04-0423. Invest Ophthalmol Vis Sci. 2004. PMID: 15452068
-
Gamma crystallins of the human eye lens.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):333-43. doi: 10.1016/j.bbagen.2015.06.007. Epub 2015 Jun 25. Biochim Biophys Acta. 2016. PMID: 26116913 Review.
-
Chordate betagamma-crystallins and the evolutionary developmental biology of the vertebrate lens.Comp Biochem Physiol B Biochem Mol Biol. 2007 Jul;147(3):347-57. doi: 10.1016/j.cbpb.2007.03.014. Epub 2007 Apr 12. Comp Biochem Physiol B Biochem Mol Biol. 2007. PMID: 17493858 Review.
Cited by
-
Crystal Structure of Chicken γS-Crystallin Reveals Lattice Contacts with Implications for Function in the Lens and the Evolution of the βγ-Crystallins.Structure. 2017 Jul 5;25(7):1068-1078.e2. doi: 10.1016/j.str.2017.05.015. Epub 2017 Jun 22. Structure. 2017. PMID: 28648607 Free PMC article.
-
Acquired Disorder and Asymmetry in a Domain-Swapped Model for γ-Crystallin Aggregation.J Mol Biol. 2022 May 15;434(9):167559. doi: 10.1016/j.jmb.2022.167559. Epub 2022 Mar 24. J Mol Biol. 2022. PMID: 35341744 Free PMC article.
-
Development of a potent embryonic chick lens model for studying congenital cataracts in vivo.Commun Biol. 2021 Mar 11;4(1):325. doi: 10.1038/s42003-021-01849-0. Commun Biol. 2021. PMID: 33707565 Free PMC article.
-
Whole genome assembly of the armored loricariid catfish Ancistrus triradiatus highlights herbivory signatures.Mol Genet Genomics. 2022 Nov;297(6):1627-1642. doi: 10.1007/s00438-022-01947-6. Epub 2022 Aug 25. Mol Genet Genomics. 2022. PMID: 36006456 Free PMC article.
-
The relationship between hard and soft tissue structures of the eye in extant lizards.J Morphol. 2022 Sep;283(9):1182-1199. doi: 10.1002/jmor.21495. Epub 2022 Jul 18. J Morphol. 2022. PMID: 35833614 Free PMC article.
References
-
- Bloemendal H, De Jong W, Jaenicke R, Lubsen NH, Slingsby C, Tardieu A. Ageing and vision: structure, stability and function of lens crystallins. Prog Biophys Mol Biol. 2004;86:407–85. - PubMed
-
- Wistow GJ, Piatigorsky J. Lens crystallins: the evolution and expression of proteins for a highly specialized tissue. Annu Rev Biochem. 1988;57:479–504. - PubMed
-
- Wistow G. Molecular Biology and Evolution of Crystallins: Gene Recruitment and Multifunctional Proteins in the Eye Lens. R.G. Landes Company; Austin, TX: 1995.
-
- Clark AR, Lubsen NH, Slingsby C. sHSP in the eye lens: Crystallin mutations, cataract and proteostasis. Int J Biochem Cell Biol. 2012;44:1687–97. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

