Control of Oxidative Stress and Inflammation in Sickle Cell Disease with the Nrf2 Activator Dimethyl Fumarate
- PMID: 26914345
- PMCID: PMC5421647
- DOI: 10.1089/ars.2015.6571
Control of Oxidative Stress and Inflammation in Sickle Cell Disease with the Nrf2 Activator Dimethyl Fumarate
Abstract
Aims: Heme derived from hemolysis is pro-oxidative and proinflammatory and promotes vaso-occlusion in murine models of sickle cell disease (SCD), suggesting that enhanced detoxification of heme may be beneficial. Nuclear factor erythroid-2-related factor-2 (Nrf2) transcription pathway is the principal cellular defense system responding to pro-oxidative and proinflammatory stress. Dimethyl fumarate (DMF), a drug approved for treatment of multiple sclerosis, provides neuroprotection by activating Nrf2-responsive genes. We hypothesized that induction of Nrf2 with DMF would be beneficial in murine SCD models.
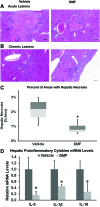
Results: DMF (30 mg/kg/day) or vehicle (0.08% methyl cellulose) was administered for 3-7 days to NY1DD and HbSS-Townes SCD mice. Vaso-occlusion, a hallmark of SCD, measured in sickle mice with dorsal skinfold chambers, was inhibited by DMF. The inhibitory effect of DMF was abrogated by the heme oxygenase-1 (HO-1) inhibitor tin protoporphyrin. DMF increased nuclear Nrf2 and cellular mRNA of Nrf2-responsive genes in livers and kidneys. DMF increased heme defenses, including HO-1, haptoglobin, hemopexin, and ferritin heavy chain, although plasma hemoglobin and heme levels were unchanged. DMF decreased markers of inflammation, including nuclear factor-kappa B phospho-p65, adhesion molecules, and toll-like receptor 4. DMF administered for 24 weeks to HbSS-Townes mice decreased hepatic necrosis, inflammatory cytokines, and irregularly shaped erythrocytes and increased hemoglobin F, but did not alter hematocrits, reticulocyte counts, lactate dehydrogenase, plasma heme, or spleen weights, indicating that the beneficial effects of DMF were not attributable to decreased hemolysis.
Innovation: These studies identify Nrf2 activation as a new therapeutic target for the treatment of SCD.
Conclusion: DMF activates Nrf2, enhances antioxidant defenses, and inhibits inflammation and vaso-occlusion in SCD mice. Antioxid. Redox Signal. 26, 748-762.
Keywords: HO-1; Nrf2; haptoglobin; hemopexin; sickle cell disease.
Conflict of interest statement
Some of the research funding to J.D.B. and G.M.V. came from Biogen, which owns the rights to Tecfidera. For all other authors, no competing financial interests exist.
Figures







References
-
- Balla G, Vercellotti GM, Muller-Eberhard U, Eaton J, and Jacob HS. Exposure of endothelial cells to free heme potentiates damage mediated by granulocytes and toxic oxygen species. Lab Invest 64: 648–655, 1991 - PubMed
-
- Barnard ML, Muller-Eberhard U, and Turrens JF. Protective role of hemopexin on heme-dependent lung oxidative stress. Biochem Biophys Res Commun 192: 82–87, 1993 - PubMed
-
- Bean CJ, Boulet SL, Ellingsen D, Pyle ME, Barron-Casella EA, Casella JF, Payne AB, Driggers J, Trau HA, Yang G, Jones K, Ofori-Acquah SF, Hooper WC, and DeBaun MR. Heme oxygenase-1 gene promoter polymorphism is associated with reduced incidence of acute chest syndrome among children with sickle cell disease. Blood 120: 3822–3828, 2012 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

