Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer
- PMID: 26915293
- PMCID: PMC4987730
- DOI: 10.1038/cdd.2016.11
Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer
Abstract
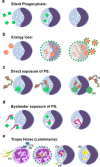
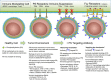
Apoptosis is an evolutionarily conserved and tightly regulated cell death modality. It serves important roles in physiology by sculpting complex tissues during embryogenesis and by removing effete cells that have reached advanced age or whose genomes have been irreparably damaged. Apoptosis culminates in the rapid and decisive removal of cell corpses by efferocytosis, a term used to distinguish the engulfment of apoptotic cells from other phagocytic processes. Over the past decades, the molecular and cell biological events associated with efferocytosis have been rigorously studied, and many eat-me signals and receptors have been identified. The externalization of phosphatidylserine (PS) is arguably the most emblematic eat-me signal that is in turn bound by a large number of serum proteins and opsonins that facilitate efferocytosis. Under physiological conditions, externalized PS functions as a dominant and evolutionarily conserved immunosuppressive signal that promotes tolerance and prevents local and systemic immune activation. Pathologically, the innate immunosuppressive effect of externalized PS has been hijacked by numerous viruses, microorganisms, and parasites to facilitate infection, and in many cases, establish infection latency. PS is also profoundly dysregulated in the tumor microenvironment and antagonizes the development of tumor immunity. In this review, we discuss the biology of PS with respect to its role as a global immunosuppressive signal and how PS is exploited to drive diverse pathological processes such as infection and cancer. Finally, we outline the rationale that agents targeting PS could have significant value in cancer and infectious disease therapeutics.
Conflict of interest statement
JC, JTH, BF, and CE are paid employees of Peregrine Pharmaceuticals. RAB receives funding from and is a consultant for Peregrine Pharmaceuticals. MH wants to disclose the involvement in Patent Application EP1356818 A3 filed at the European Patent Office in 1995 and entitled ‘Drug, in particular for modulating the immunological response for the control of viruses, tumors, bacteria, and parasites'. This patent application has already expired. Peregrine Pharmaceuticals, Inc., is the owner or exclusive licensee of hundreds of unexpired worldwide patents broadly covering compositions that target PS and various methods of use, including to treat cancer and viral infections. The remaining authors declare no conflict of interests.
Figures





References
-
- Leventis PA, Grinstein S. The distribution and function of phosphatidylserine in cellular membranes. Annu Rev Biophys 2010; 39: 407–427. - PubMed
-
- Schroit AJ, Madsen JW, Tanaka Y. In vivo recognition and clearance of red blood cells containing phosphatidylserine in their plasma membranes. J Biol Chem 1985; 260: 5131–5138. - PubMed
-
- Stone SJ, Vance JE. Phosphatidylserine synthase-1 and -2 are localized to mitochondria-associated membranes. J Biol Chem 2000; 275: 34534–34540. - PubMed
-
- Arikketh D, Nelson R, Vance JE. Defining the importance of phosphatidylserine synthase-1 (PSS1): unexpected viability of PSS1-deficient mice. J Biol Chem 2008; 283: 12888–12897. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

