The therapeutic potential of genome editing for β-thalassemia
- PMID: 26918126
- PMCID: PMC4753996
- DOI: 10.12688/f1000research.7087.1
The therapeutic potential of genome editing for β-thalassemia
Abstract
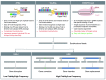
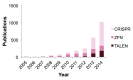
The rapid advances in the field of genome editing using targeted endonucleases have called considerable attention to the potential of this technology for human gene therapy. Targeted correction of disease-causing mutations could ensure lifelong, tissue-specific expression of the relevant gene, thereby alleviating or resolving a specific disease phenotype. In this review, we aim to explore the potential of this technology for the therapy of β-thalassemia. This blood disorder is caused by mutations in the gene encoding the β-globin chain of hemoglobin, leading to severe anemia in affected patients. Curative allogeneic bone marrow transplantation is available only to a small subset of patients, leaving the majority of patients dependent on regular blood transfusions and iron chelation therapy. The transfer of gene-corrected autologous hematopoietic stem cells could provide a therapeutic alternative, as recent results from gene therapy trials using a lentiviral gene addition approach have demonstrated. Genome editing has the potential to further advance this approach as it eliminates the need for semi-randomly integrating viral vectors and their associated risk of insertional mutagenesis. In the following pages we will highlight the advantages and risks of genome editing compared to standard therapy for β-thalassemia and elaborate on lessons learned from recent gene therapy trials.
Keywords: gene therapy; genome; thalassemia.
Conflict of interest statement
No competing interests were disclosed.
Figures
References
-
- Lanzkowsky P: Manual of pediatric hematology and oncology.2011;5 Reference Source
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

