Transient Receptor Potential Melastatin 7 Cation Channel Kinase: New Player in Angiotensin II-Induced Hypertension
- PMID: 26928801
- PMCID: PMC4786473
- DOI: 10.1161/HYPERTENSIONAHA.115.07021
Transient Receptor Potential Melastatin 7 Cation Channel Kinase: New Player in Angiotensin II-Induced Hypertension
Abstract
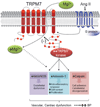
Transient receptor potential melastatin 7 (TRPM7) is a bifunctional protein comprising a magnesium (Mg(2+))/cation channel and a kinase domain. We previously demonstrated that vasoactive agents regulate vascular TRPM7. Whether TRPM7 plays a role in the pathophysiology of hypertension and associated cardiovascular dysfunction is unknown. We studied TRPM7 kinase-deficient mice (TRPM7Δkinase; heterozygous for TRPM7 kinase) and wild-type (WT) mice infused with angiotensin II (Ang II; 400 ng/kg per minute, 4 weeks). TRPM7 kinase expression was lower in heart and aorta from TRPM7Δkinase versus WT mice, effects that were further reduced by Ang II infusion. Plasma Mg(2+) was lower in TRPM7Δkinase versus WT mice in basal and stimulated conditions. Ang II increased blood pressure in both strains with exaggerated responses in TRPM7Δkinase versus WT groups (P<0.05). Acetylcholine-induced vasorelaxation was reduced in Ang II-infused TRPM7Δkinase mice, an effect associated with Akt and endothelial nitric oxide synthase downregulation. Vascular cell adhesion molecule-1 expression was increased in Ang II-infused TRPM7 kinase-deficient mice. TRPM7 kinase targets, calpain, and annexin-1, were activated by Ang II in WT but not in TRPM7Δkinase mice. Echocardiographic and histopathologic analysis demonstrated cardiac hypertrophy and left ventricular dysfunction in Ang II-treated groups. In TRPM7 kinase-deficient mice, Ang II-induced cardiac functional and structural effects were amplified compared with WT counterparts. Our data demonstrate that in TRPM7Δkinase mice, Ang II-induced hypertension is exaggerated, cardiac remodeling and left ventricular dysfunction are amplified, and endothelial function is impaired. These processes are associated with hypomagnesemia, blunted TRPM7 kinase expression/signaling, endothelial nitric oxide synthase downregulation, and proinflammatory vascular responses. Our findings identify TRPM7 kinase as a novel player in Ang II-induced hypertension and associated vascular and target organ damage.
Keywords: blood pressure; downregulation; hypertension; magnesium; signal transduction.
© 2016 American Heart Association, Inc.
Figures







References
-
- Bates-Withers C, Sah R, Clapham DE. TRPM7, the Mg(2+) inhibited channel and kinase. Adv Exp Med Biol. 2011;704:173–183. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

