Nitric Oxide Exerts Basal and Insulin-Dependent Anorexigenic Actions in POMC Hypothalamic Neurons
- PMID: 26930171
- PMCID: PMC5414647
- DOI: 10.1210/me.2015-1275
Nitric Oxide Exerts Basal and Insulin-Dependent Anorexigenic Actions in POMC Hypothalamic Neurons
Abstract
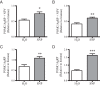
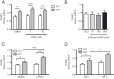
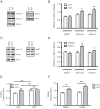
The arcuate nucleus of the hypothalamus represents a key center for the control of appetite and feeding through the regulation of 2 key neuronal populations, notably agouti-related peptide/neuropeptide Y and proopimelanocortin (POMC)/cocaine- and amphetamine-regulated transcript neurons. Altered regulation of these neuronal networks, in particular the dysfunction of POMC neurons upon high-fat consumption, is a major pathogenic mechanism involved in the development of obesity and type 2 diabetes mellitus. Efforts are underway to preserve the integrity or enhance the functionality of POMC neurons in order to prevent or treat these metabolic diseases. Here, we report for the first time that the nitric oxide (NO(-)) donor, sodium nitroprusside (SNP) mediates anorexigenic actions in both hypothalamic tissue and hypothalamic-derived cell models by mediating the up-regulation of POMC levels. SNP increased POMC mRNA in a dose-dependent manner and enhanced α-melanocortin-secreting hormone production and secretion in mHypoA-POMC/GFP-2 cells. SNP also enhanced insulin-driven POMC expression likely by inhibiting the deacetylase activity of sirtuin 1. Furthermore, SNP enhanced insulin-dependent POMC expression, likely by reducing the transcriptional repression of Foxo1 on the POMC gene. Prolonged SNP exposure prevented the development of insulin resistance. Taken together, the NO(-) donor SNP enhances the anorexigenic potential of POMC neurons by promoting its transcriptional expression independent and in cooperation with insulin. Thus, increasing cellular NO(-) levels represents a hormone-independent method of promoting anorexigenic output from the existing POMC neuronal populations and may be advantageous in the fight against these prevalent disorders.
Figures





References
-
- Belgardt BF, Brüning JC. CNS leptin and insulin action in the control of energy homeostasis. Ann NY Acad Sci. 2010;1212:97–113. - PubMed
-
- Brüning JC, Winnay J, Bonner-Weir S, Taylor SI, Accili D, Kahn CR. Development of a novel polygenic model of NIDDM in mice heterozygous for IR and IRS-1 null alleles. Cell. 1997;88:561–572. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

