Heme oxygenase-1 nuclear translocation regulates bortezomibinduced cytotoxicity and mediates genomic instability in myeloma cells
- PMID: 26930712
- PMCID: PMC5045362
- DOI: 10.18632/oncotarget.7563
Heme oxygenase-1 nuclear translocation regulates bortezomibinduced cytotoxicity and mediates genomic instability in myeloma cells
Abstract
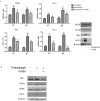
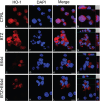
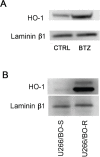
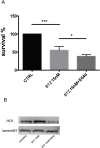
Multiple myeloma (MM) is a clonal B-cell malignancy characterized by an accumulation of clonal plasma cells in the bone marrow leading to bone destruction and bone marrow failure. Several molecular mechanisms underlie chemoresistance among which heme oxygenase-1 (HO-1) could play a major role. The aim of the present research was to evaluate the impact of HO-1 in MM following bortezomib (BTZ) treatment and how HO-1 is implicated in the mechanisms of chemoresistance. MM cells were treated for 24h with BTZ (15 nM), a boronic acid dipeptide inhibitor of the 26S proteasome used in the treatment of patients with MM as first-line therapy. We evaluated cell viability, reactive oxygen species (ROS) formation, endoplasmic reticulum (ER) stress, HO-1 expression and compartmentalization and cellular genetic instability. Results showed that BTZ significantly reduced cell viability in different MM cell lines and induced ER-stress and ROS formation. Concomitantly, we observed a significant overexpression of both HO-1 gene and protein levels. This effect was abolished by concomitant treatment with 4-phenybutirric acid, a molecular chaperone, which is known to reduce ER-stress. Surprisingly, inhibition of HO activity with SnMP (10μM) failed to increase BTZ sensitivity in MM cells whereas inhibition of HO-1 nuclear translocation by E64d, a cysteine protease inhibitor, increased sensitivity to BTZ and decreased genetic instability as measured by cytokinesis-block micronucleus assay. In conclusion, our data suggest that BTZ sensitivity depends on HO-1 nuclear compartmentalization and not on its enzymatic activity and this finding may represent an important tool to overcome BTZ chemoresistance in MM patients.
Keywords: endoplasmic reticulum stress; genomic instability; heme oxygenase; multiple myeloma; oxidative stress.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article.
Figures



References
-
- Morgan GJ, Walker BA, Davies FE. The genetic architecture of multiple myeloma. Nat Rev Cancer. 2012;12:335–348. - PubMed
-
- Masella R, Santangelo C, D'Archivio M, Li Volti G, Giovannini C, Galvano F. Protocatechuic acid and human disease prevention: biological activities and molecular mechanisms. Curr Med Chem. 2012;19:2901–2917. - PubMed
-
- Meister S, Schubert U, Neubert K, Herrmann K, Burger R, Gramatzki M, Hahn S, Schreiber S, Wilhelm S, Herrmann M, Jack HM, Voll RE. Extensive immunoglobulin production sensitizes myeloma cells for proteasome inhibition. Cancer Res. 2007;67:1783–1792. - PubMed
-
- Nakamura M, Gotoh T, Okuno Y, Tatetsu H, Sonoki T, Uneda S, Mori M, Mitsuya H, Hata H. Activation of the endoplasmic reticulum stress pathway is associated with survival of myeloma cells. Leuk Lymphoma. 2006;47:531–539. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

