Stem Cell-Derived Immature Human Dorsal Root Ganglia Neurons to Identify Peripheral Neurotoxicants
- PMID: 26933043
- PMCID: PMC4798731
- DOI: 10.5966/sctm.2015-0108
Stem Cell-Derived Immature Human Dorsal Root Ganglia Neurons to Identify Peripheral Neurotoxicants
Abstract
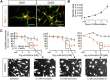
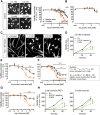
Safety sciences and the identification of chemical hazards have been seen as one of the most immediate practical applications of human pluripotent stem cell technology. Protocols for the generation of many desirable human cell types have been developed, but optimization of neuronal models for toxicological use has been astonishingly slow, and the wide, clinically important field of peripheral neurotoxicity is still largely unexplored. A two-step protocol to generate large lots of identical peripheral human neuronal precursors was characterized and adapted to the measurement of peripheral neurotoxicity. High content imaging allowed an unbiased assessment of cell morphology and viability. The computational quantification of neurite growth as a functional parameter highly sensitive to disturbances by toxicants was used as an endpoint reflecting specific neurotoxicity. The differentiation of cells toward dorsal root ganglia neurons was tracked in relation to a large background data set based on gene expression microarrays. On this basis, a peripheral neurotoxicity (PeriTox) test was developed as a first toxicological assay that harnesses the potential of human pluripotent stem cells to generate cell types/tissues that are not otherwise available for the prediction of human systemic organ toxicity. Testing of more than 30 chemicals showed that human neurotoxicants and neurite growth enhancers were correctly identified. Various classes of chemotherapeutic agents causing human peripheral neuropathies were identified, and they were missed when tested on human central neurons. The PeriTox test we established shows the potential of human stem cells for clinically relevant safety testing of drugs in use and of new emerging candidates.
Significance: The generation of human cells from pluripotent stem cells has aroused great hopes in biomedical research and safety sciences. Neurotoxicity testing is a particularly important application for stem cell-derived somatic cells, as human neurons are hardly available otherwise. Also, peripheral neurotoxicity has become of major concern in drug development for chemotherapy. The first neurotoxicity test method was established based on human pluripotent stem cell-derived peripheral neurons. The strategies exemplified in the present study of reproducible cell generation, cell function-based test system establishment, and assay validation provide the basis for a drug safety assessment on cells not available otherwise.
Keywords: Human pluripotent stem cells; In vitro drug and toxicity testing; Neural differentiation; Peripheral neuropathies.
©AlphaMed Press.
Figures





Similar articles
-
Evaluation of a human neurite growth assay as specific screen for developmental neurotoxicants.Arch Toxicol. 2013 Dec;87(12):2215-31. doi: 10.1007/s00204-013-1072-y. Epub 2013 May 14. Arch Toxicol. 2013. PMID: 23670202
-
Human Pluripotent Stem Cells as Tools for Predicting Developmental Neural Toxicity of Chemicals: Strategies, Applications, and Challenges.Stem Cells Dev. 2019 Jun 15;28(12):755-768. doi: 10.1089/scd.2019.0007. Epub 2019 May 24. Stem Cells Dev. 2019. PMID: 30990109 Review.
-
Towards in vitro DT/DNT testing: Assaying chemical susceptibility in early differentiating NT2 cells.Toxicology. 2015 Dec 2;338:69-76. doi: 10.1016/j.tox.2015.10.007. Epub 2015 Oct 20. Toxicology. 2015. PMID: 26498558
-
Human-Induced Pluripotent Stem Cell-Derived Neural Organoids as a Novel In Vitro Platform for Developmental Neurotoxicity Assessment.Int J Mol Sci. 2024 Nov 21;25(23):12523. doi: 10.3390/ijms252312523. Int J Mol Sci. 2024. PMID: 39684235 Free PMC article.
-
In vitro acute and developmental neurotoxicity screening: an overview of cellular platforms and high-throughput technical possibilities.Arch Toxicol. 2017 Jan;91(1):1-33. doi: 10.1007/s00204-016-1805-9. Epub 2016 Aug 4. Arch Toxicol. 2017. PMID: 27492622 Review.
Cited by
-
Time and space-resolved quantification of plasma membrane sialylation for measurements of cell function and neurotoxicity.Arch Toxicol. 2020 Feb;94(2):449-467. doi: 10.1007/s00204-019-02642-z. Epub 2019 Dec 11. Arch Toxicol. 2020. PMID: 31828357
-
Human iPSC-derived trigeminal neurons lack constitutive TLR3-dependent immunity that protects cortical neurons from HSV-1 infection.Proc Natl Acad Sci U S A. 2018 Sep 11;115(37):E8775-E8782. doi: 10.1073/pnas.1809853115. Epub 2018 Aug 28. Proc Natl Acad Sci U S A. 2018. PMID: 30154162 Free PMC article.
-
A Comparative Review of Chemotherapy-Induced Peripheral Neuropathy in In Vivo and In Vitro Models.Toxicol Pathol. 2020 Jan;48(1):190-201. doi: 10.1177/0192623319861937. Epub 2019 Jul 22. Toxicol Pathol. 2020. PMID: 31331249 Free PMC article. Review.
-
Specific Attenuation of Purinergic Signaling during Bortezomib-Induced Peripheral Neuropathy In Vitro.Int J Mol Sci. 2022 Mar 29;23(7):3734. doi: 10.3390/ijms23073734. Int J Mol Sci. 2022. PMID: 35409095 Free PMC article.
-
Modeling chemotherapy induced peripheral neuropathy (CIPN) in vitro: Prospects and limitations.Exp Neurol. 2020 Apr;326:113140. doi: 10.1016/j.expneurol.2019.113140. Epub 2019 Dec 5. Exp Neurol. 2020. PMID: 31812556 Free PMC article. Review.
References
-
- Spencer PS, Schaumburg HH. Experimental and Clinical Neurotoxicology. New York: Oxford University Press; 2000.
-
- Kuegler PB, Zimmer B, Waldmann T, et al. Markers of murine embryonic and neural stem cells, neurons and astrocytes: Reference points for developmental neurotoxicity testing. ALTEX. 2010;27:17–42. - PubMed
-
- Bouvier d’Yvoire M, Bremer S, Casati S, et al. ECVAM and new technologies for toxicity testing. Adv Exp Med Biol. 2012;745:154–180. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

