EspP, an Extracellular Serine Protease from Enterohemorrhagic E. coli, Reduces Coagulation Factor Activities, Reduces Clot Strength, and Promotes Clot Lysis
- PMID: 26934472
- PMCID: PMC4775034
- DOI: 10.1371/journal.pone.0149830
EspP, an Extracellular Serine Protease from Enterohemorrhagic E. coli, Reduces Coagulation Factor Activities, Reduces Clot Strength, and Promotes Clot Lysis
Abstract
Background: EspP (E. coli secreted serine protease, large plasmid encoded) is an extracellular serine protease produced by enterohemorrhagic E. coli (EHEC) O157:H7, a causative agent of diarrhea-associated Hemolytic Uremic Syndrome (D+HUS). The mechanism by which EHEC induces D+HUS has not been fully elucidated.
Objectives: We investigated the effects of EspP on clot formation and lysis in human blood.
Methods: Human whole blood and plasma were incubated with EspP(WT )at various concentrations and sampled at various time points. Thrombin time (TT), prothrombin time (PT), and activated partial thromboplastin time (aPTT), coagulation factor activities, and thrombelastgraphy (TEG) were measured.
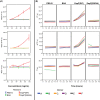
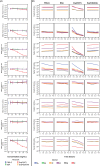
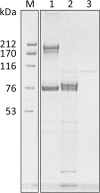
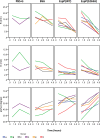
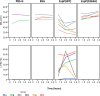
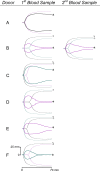
Results and conclusions: Human whole blood or plasma incubated with EspP(WT) was found to have prolonged PT, aPTT, and TT. Furthermore, human whole blood or plasma incubated with EspP(WT) had reduced activities of coagulation factors V, VII, VIII, and XII, as well as prothrombin. EspP did not alter the activities of coagulation factors IX, X, or XI. When analyzed by whole blood TEG, EspP decreased the maximum amplitude of the clot, and increased the clot lysis. Our results indicate that EspP alters hemostasis in vitro by decreasing the activities of coagulation factors V, VII, VIII, and XII, and of prothrombin, by reducing the clot strength and accelerating fibrinolysis, and provide further evidence of a functional role for this protease in the virulence of EHEC and the development of D+HUS.
Conflict of interest statement
Figures
Similar articles
-
Subtypes of the plasmid-encoded serine protease EspP in Shiga toxin-producing Escherichia coli: distribution, secretion, and proteolytic activity.Appl Environ Microbiol. 2007 Oct;73(20):6351-9. doi: 10.1128/AEM.00920-07. Epub 2007 Aug 17. Appl Environ Microbiol. 2007. PMID: 17704265 Free PMC article.
-
EspP, a serine protease of enterohemorrhagic Escherichia coli, impairs complement activation by cleaving complement factors C3/C3b and C5.Infect Immun. 2010 Oct;78(10):4294-301. doi: 10.1128/IAI.00488-10. Epub 2010 Jul 19. Infect Immun. 2010. PMID: 20643852 Free PMC article.
-
EspP, a novel extracellular serine protease of enterohaemorrhagic Escherichia coli O157:H7 cleaves human coagulation factor V.Mol Microbiol. 1997 May;24(4):767-78. doi: 10.1046/j.1365-2958.1997.3871751.x. Mol Microbiol. 1997. PMID: 9194704
-
Prevalence, biogenesis, and functionality of the serine protease autotransporter EspP.Toxins (Basel). 2012 Dec 28;5(1):25-48. doi: 10.3390/toxins5010025. Toxins (Basel). 2012. PMID: 23274272 Free PMC article. Review.
-
Theory of blood coagulation in disseminated intravascular coagulation (D. I. C.).Folia Haematol Int Mag Klin Morphol Blutforsch. 1972;97(1):5-19. Folia Haematol Int Mag Klin Morphol Blutforsch. 1972. PMID: 4114585 Review. No abstract available.
Cited by
-
IgG Binds Escherichia coli Serine Protease EspP and Protects Mice From E. coli O157:H7 Infection.Front Immunol. 2022 Feb 18;13:807959. doi: 10.3389/fimmu.2022.807959. eCollection 2022. Front Immunol. 2022. PMID: 35250980 Free PMC article.
References
-
- Karch H (2001) The role of virulence factors in enterohemorrhagic Escherichia coli (EHEC)-associated hemolytic-uremic syndrome. Semin Thromb Hemost 27: 207–213. - PubMed
-
- Brunder W, Schmidt H, Karch H (1997) EspP, a novel extracellular serine protease of enterohaemorrhagic Escherichia coli O157:H7 cleaves human coagulation factor V. Mol Microbiol 24: 767–778. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

