ω-3 Fatty acids for major depressive disorder in adults: an abridged Cochrane review
- PMID: 26936905
- PMCID: PMC4785331
- DOI: 10.1136/bmjopen-2015-010172
ω-3 Fatty acids for major depressive disorder in adults: an abridged Cochrane review
Erratum in
-
Correction.BMJ Open. 2017 Jan 16;7(1):e010172corr1. doi: 10.1136/bmjopen-2015-010172corr1. BMJ Open. 2017. PMID: 28093445 Free PMC article. No abstract available.
Abstract
Objective: To assess the effects of n-3 polyunsaturated fatty acids (n-3PUFAs; also known as ω-3 fatty acids) compared with comparator for major depressive disorder (MDD) in adults.
Design: Systematic review and meta-analyses.
Data sources: The Cochrane Depression, Anxiety and Neurosis Review Group's Specialised Registers (CCDANCTR) and International Trial Registries searched to May 2015. CINAHL searched to September 2013.
Inclusion criteria: a randomised controlled trial (RCT); that provided n-3PUFAs as an intervention; used a comparator; measured depressive symptomology as an outcome; and was conducted in adults with MDD.
Outcomes: Primary outcomes were depressive symptomology and adverse events.
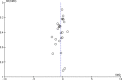
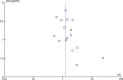
Results: 20 trials encompassing 26 relevant studies were found. For n-3PUFAs versus placebo, n-3PUFA supplementation resulted in a small-to-modest benefit for depressive symptomology: SMD=-0.32 (95% CI -0.52 to -0.12; 25 studies, 1373 participants, very low-quality evidence), but this effect is unlikely to be clinically meaningful, is very imprecise and, based on funnel plot inspection, sensitivity analyses and comparison with large well-conducted trials, is likely to be biased. Considerable evidence of heterogeneity between studies was also found, and was not explained by subgroup or sensitivity analyses. Numbers of individuals experiencing adverse events were similar in intervention and placebo groups (OR=1.24, 95% CI 0.95 to 1.62; 19 studies, 1207 participants; very low-quality evidence). For n-3PUFAs versus antidepressants, no differences were found between treatments in depressive symptomology (MD=-0.70 (95% CI -5.88 to 4.48); 1 study, 40 participants, very low-quality evidence).
Conclusions: At present, we do not have sufficient evidence to determine the effects of n-3PUFAs as a treatment for MDD. Further research in the form of adequately powered RCTs is needed.
Keywords: EPIDEMIOLOGY; NUTRITION & DIETETICS; THERAPEUTICS.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/
Figures




References
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th edn Washington DC: American Psychiatric Association, 2013.
-
- World Health Organization. Global Burden of Disease. http://www.who.int/healthinfo/global_burden_disease/gbd/en/index.html 1 Febuary 2014.
-
- Haag M. Essential fatty acids and the brain. Can J Psychiatry 2003;48:195–203. - PubMed
-
- Ma J, Folsom AR, Eckfeldt JH et al. Short- and long-term repeatability of fatty acid composition of human plasma phospholipids and cholesterol esters. The Atherosclerosis Risk in Communities (ARIC) Study Investigators. Am J Clin Nutr 1995;62:572–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
