A North American H7N3 Influenza Virus Supports Reassortment with 2009 Pandemic H1N1 and Induces Disease in Mice without Prior Adaptation
- PMID: 26937034
- PMCID: PMC4836326
- DOI: 10.1128/JVI.02761-15
A North American H7N3 Influenza Virus Supports Reassortment with 2009 Pandemic H1N1 and Induces Disease in Mice without Prior Adaptation
Abstract
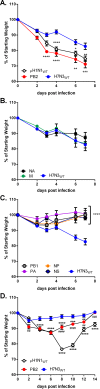
Reassortment between H5 or H9 subtype avian and mammalian influenza A viruses (IAV) can generate a novel virus that causes disease and transmits between mammals. Such information is currently not available for H7 subtype viruses. We evaluated the ability of a low-pathogenicity North American avian H7N3 virus (A/shorebird/Delaware/22/2006) to reassort with mammalian or avian viruses using a plasmid-based competition assay. In addition to genome segments derived from an avian H7N9 virus, the H7N3 virus reassorted efficiently with the PB2, NA, and M segments from the 2009 pandemic H1N1 (PH1N1) virus.In vitro and in vivo evaluation of the H7N3:PH1N1 (7 + 1) reassortant viruses revealed that the PB2, NA, or M segments from PH1N1 largely do not attenuate the H7N3 virus, whereas the PB1, PA, NP, or NS genome segments from PH1N1 do. Additionally, we assessed the functionality of the H7N3:PH1N1 7 + 1 reassortant viruses by measuring the inflammatory response in vivo We found that infection with wild-type H7N3 resulted in increased inflammatory cytokine production relative to that seen with the PH1N1 strain and that the increase was further exacerbated by substitution of PH1N1 PB2 but not NA or M. Finally, we assessed if any adaptations occurred in the individually substituted segments after in vivo inoculation and found no mutations, suggesting that PH1N1 PB2, NA, and M are genetically stable in the background of this H7N3 virus. Taking the data together, we demonstrate that a North American avian H7N3 IAV is genetically and functionally compatible with multiple gene segments from the 2009 pandemic influenza virus strain without prior adaptation.
Importance: The 2009 pandemic H1N1 virus continues to circulate and reassort with other influenza viruses, creating novel viruses with increased replication and transmission potential in humans. Previous studies have found that this virus can also reassort with H5N1 and H9N2 avian influenza viruses. We now show that several genome segments of the 2009 H1N1 virus are also highly compatible with a low-pathogenicity avian H7N3 virus and that these reassortant viruses are stable and not attenuated in an animal model. These results highlight the potential for reassortment of H1N1 viruses with avian influenza virus and emphasize the need for continued surveillance of influenza viruses in areas of cocirculation between avian, human, and swine viruses.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures



Similar articles
-
Seasonal H3N2 and 2009 Pandemic H1N1 Influenza A Viruses Reassort Efficiently but Produce Attenuated Progeny.J Virol. 2017 Aug 10;91(17):e00830-17. doi: 10.1128/JVI.00830-17. Print 2017 Sep 1. J Virol. 2017. PMID: 28637755 Free PMC article.
-
Differences in transmissibility and pathogenicity of reassortants between H9N2 and 2009 pandemic H1N1 influenza A viruses from humans and swine.Arch Virol. 2014 Jul;159(7):1743-54. doi: 10.1007/s00705-014-2009-3. Epub 2014 Feb 9. Arch Virol. 2014. PMID: 24510170
-
The M segment of the 2009 pandemic influenza virus confers increased neuraminidase activity, filamentous morphology, and efficient contact transmissibility to A/Puerto Rico/8/1934-based reassortant viruses.J Virol. 2014 Apr;88(7):3802-14. doi: 10.1128/JVI.03607-13. Epub 2014 Jan 15. J Virol. 2014. PMID: 24429367 Free PMC article.
-
Isolation and genetic characterization of avian-like H1N1 and novel ressortant H1N2 influenza viruses from pigs in China.Biochem Biophys Res Commun. 2009 Aug 21;386(2):278-83. doi: 10.1016/j.bbrc.2009.05.056. Epub 2009 May 19. Biochem Biophys Res Commun. 2009. PMID: 19460353 Review.
-
Virulence of pandemic (H1N1) 2009 influenza A polymerase reassortant viruses.Virulence. 2011 Sep-Oct;2(5):422-6. doi: 10.4161/viru.2.5.17267. Epub 2011 Sep 1. Virulence. 2011. PMID: 21921678 Review.
Cited by
-
mRNA induced expression of human angiotensin-converting enzyme 2 in mice for the study of the adaptive immune response to severe acute respiratory syndrome coronavirus 2.PLoS Pathog. 2020 Dec 16;16(12):e1009163. doi: 10.1371/journal.ppat.1009163. eCollection 2020 Dec. PLoS Pathog. 2020. PMID: 33326500 Free PMC article.
-
Nucleotide resolution mapping of influenza A virus nucleoprotein-RNA interactions reveals RNA features required for replication.Nat Commun. 2018 Jan 31;9(1):465. doi: 10.1038/s41467-018-02886-w. Nat Commun. 2018. PMID: 29386621 Free PMC article.
-
Irg1 expression in myeloid cells prevents immunopathology during M. tuberculosis infection.J Exp Med. 2018 Apr 2;215(4):1035-1045. doi: 10.1084/jem.20180118. Epub 2018 Mar 6. J Exp Med. 2018. PMID: 29511063 Free PMC article.
-
Residues in the PB2 and PA genes contribute to the pathogenicity of avian H7N3 influenza A virus in DBA/2 mice.Virology. 2016 Jul;494:89-99. doi: 10.1016/j.virol.2016.04.013. Epub 2016 Apr 20. Virology. 2016. PMID: 27105450 Free PMC article.
-
Interferon induced protein 35 exacerbates H5N1 influenza disease through the expression of IL-12p40 homodimer.PLoS Pathog. 2018 Apr 26;14(4):e1007001. doi: 10.1371/journal.ppat.1007001. eCollection 2018 Apr. PLoS Pathog. 2018. PMID: 29698474 Free PMC article.
References
-
- Yu H, Cowling BJ, Feng L, Lau EHY, Liao Q, Tsang TK, Peng Z, Wu P, Liu F, Fang VJ, Zhang H, Li M, Zeng L, Xu Z, Li Z, Luo H, Li Q, Feng Z, Cao B, Yang W, Wu JT, Wang Y, Leung GM. 2013. Human infection with avian influenza A H7N9 virus: an assessment of clinical severity. Lancet 382:138–145. doi: 10.1016/S0140-6736(13)61207-6. - DOI - PMC - PubMed
-
- Itoh Y, Shinya K, Kiso M, Watanabe T, Sakoda Y, Hatta M, Muramoto Y, Tamura D, Sakai-Tagawa Y, Noda T, Sakabe S, Imai M, Hatta Y, Watanabe S, Li C, Yamada S, Fujii K, Murakami S, Imai H, Kakugawa S, Ito M, Takano R, Iwatsuki-Horimoto K, Shimojima M, Horimoto T, Goto H, Takahashi K, Makino A, Ishigaki H, Nakayama M, Okamatsu M, Takahashi K, Warshauer D, Shult PA, Saito R, Suzuki H, Furuta Y, Yamashita M, Mitamura K, Nakano K, Nakamura M, Brockman-Schneider R, Mitamura H, Yamazaki M, Sugaya N, Suresh M, Ozawa M, Neumann G, Gern J, Kida H, Ogasawara K, Kawaoka Y. 2009. In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature 460:1021–1025. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

