Combined Effects of GSTM1 Null Allele and APOL1 Renal Risk Alleles in CKD Progression in the African American Study of Kidney Disease and Hypertension Trial
- PMID: 26940095
- PMCID: PMC5042657
- DOI: 10.1681/ASN.2015050487
Combined Effects of GSTM1 Null Allele and APOL1 Renal Risk Alleles in CKD Progression in the African American Study of Kidney Disease and Hypertension Trial
Abstract
Apolipoprotein L-1 (APOL1) high-risk alleles and the glutathione-S-transferase-μ1 (GSTM1) null allele have been shown separately to associate with CKD progression in the African American Study of Kidney Disease and Hypertension (AASK) trial participants. Here, we determined combined effects of GSTM1 null and APOL1 high-risk alleles on clinical outcomes in 682 AASK participants who were classified into four groups by GSTM1 null or active genotype and APOL1 high- or low-risk genotype. We assessed survival differences among these groups by log-rank test and Cox regression adjusted for important clinical variables for time to GFR event (change in GFR of 50% or 25-ml/min per 1.73 m2 decline), incident ESRD, death, or composite outcomes. The groups differed significantly in event-free survival for incident ESRD and composite outcomes (P≤0.001 by log-rank test). Compared with the reference GSTM1 active/APOL1 low-risk group, other groups had these hazard ratios for the composite outcome of incident ESRD and change in GFR: GSTM1 active/APOL1 high-risk hazard ratio, 2.13; 95% confidence interval, 0.76 to 5.90 (P=0.15); GSTM1 null/APOL1 low-risk hazard ratio, 2.05; 95% confidence interval, 1.08 to 3.88 (P=0.03); and GSTM1 null/APOL1 high-risk hazard ratio, 3.0; 95% confidence interval, 1.51 to 5.96 (P=0.002). In conclusion, GSTM1 null and APOL1 high-risk alleles deleteriously affect CKD progression among blacks with hypertension, and subjects with both GSTM1 null and APOL1 high-risk genotypes had highest risk of adverse renal outcomes. Larger cohorts are needed to fully explore interactions of GSTM1 and APOL1 genotypes in other subgroups.
Keywords: AASK (African American Study of Kidney Disease and Hypertension); genetic renal disease; kidney disease.
Copyright © 2016 by the American Society of Nephrology.
Figures
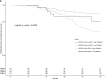


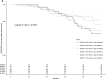


References
-
- US Renal Data System : USRDS 2011 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States, Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2011
-
- Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI, Bowden DW, Langefeld CD, Oleksyk TK, Uscinski Knob AL, Bernhardy AJ, Hicks PJ, Nelson GW, Vanhollebeke B, Winkler CA, Kopp JB, Pays E, Pollak MR: Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329: 841–845, 2010 - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

