Administration of nintedanib after discontinuation for acute exacerbation of idiopathic pulmonary fibrosis: a case report
- PMID: 26940352
- PMCID: PMC4778330
- DOI: 10.1186/s12890-016-0201-9
Administration of nintedanib after discontinuation for acute exacerbation of idiopathic pulmonary fibrosis: a case report
Abstract
Background: Nintedanib is a multi-target receptor tyrosine kinase inhibitor. In two recent randomized phase 3 trials (INPULSIS™-1 and -2), it has been shown to slow the disease progression of idiopathic pulmonary fibrosis (IPF) by reducing the decline in the forced vital capacity (FVC). Although the INPULSIS™ trials indicate that nintedanib may serve to prevent acute exacerbations or delay the time to the first acute exacerbation, a certain number of IPF patients develop acute exacerbations while receiving nintedanib. However, there has been no report on the readministration of nintedanib in IPF patients who develop acute exacerbations during initial treatment with nintedanib.
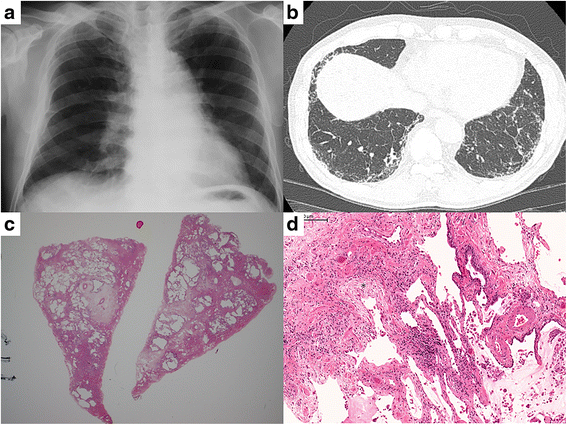
Case presentation: A 64-year-old man with IPF had nintedanib added to his ongoing pirfenidone therapy. He developed dyspnea after 65 days and presented with hypoxemia after 68 days. At presentation, chest computed tomography showed newly developed diffuse ground glass opacities with the pre-existing subpleural reticular shadows. Because of the absence of infection or other potential causative factors, we diagnosed an acute exacerbation of IPF. Nintedanib was temporarily discontinued and the acute exacerbation was successfully managed with intensive treatment. We re-initiated nintedanib 30 days after cessation, which helped stabilize his FVC for 8 months. Nintedanib was safely continued for 28 months until he died of a bacterial infection.
Conclusion: To the best of our our knowledge, this is the first reported case of an acute exacerbation of IPF during nintedanib treatment, wherein nintedanib was safely and successfully restarted after treatment of the acute exacerbation. Our case indicates that nintedanib can be safely resumed and a desired effect on FVC can be obtained, even in IPF patients who develop acute exacerbations. However, we recommend close monitoring and appropriate measures until the long-term safety profile is clarified.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

