Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for colorectal cancer: survival outcomes and patient selection
- PMID: 26941985
- PMCID: PMC4754309
- DOI: 10.3978/j.issn.2078-6891.2015.114
Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for colorectal cancer: survival outcomes and patient selection
Abstract
Background: Chemotherapy hyperthermic intraperitoneal chemotherapy (HIPEC) is playing an ever increasing role in the management of colorectal cancer (CRC) with peritoneal metastases (PM) as results approach those of surgical resection of liver metastases. Selection criteria for treatment type, sequence and timing of currently available therapies remain ill-defined.
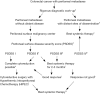
Methods: We review the current published literature analyzing outcomes by treatments with surgery, systemic chemotherapy, cytoreductive surgery (CRS) and HIPEC, and ongoing clinical trials. A clinical pathway that incorporates all currently available therapies, determining the timing and sequence of such therapies was constructed.
Results: Most of the literature on outcome data comes from studies reporting the results of CRS and HIPEC with large series showing a median survival of 32-47 months. Meanwhile, the vast majority of patients, over 90% in the United States, are being treated with palliative systemic therapies following the NCCN guidelines.
Conclusions: Cooperation between medical and surgical oncologists represents an unmet need in oncology when it comes to patients with CRC with PM. The presented clinical pathway constitutes a feasible and much needed first step to start this cooperation.
Keywords: Cytoreductive surgery (CRS); colorectal cancer (CRC); hyperthermic intraperitoneal chemotherapy (HIPEC); peritoneal carcinomatosis.
Conflict of interest statement
Figures
Similar articles
-
Management of colorectal cancer patients at high risk of peritoneal metastases.J BUON. 2015 May;20 Suppl 1:S71-9. J BUON. 2015. PMID: 26051336
-
Trends in cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for the treatment of synchronous peritoneal carcinomatosis of colorectal origin in the Netherlands.Eur J Surg Oncol. 2015 Apr;41(4):466-71. doi: 10.1016/j.ejso.2015.01.018. Epub 2015 Jan 29. Eur J Surg Oncol. 2015. PMID: 25680955
-
Hepatectomy for metachronous colorectal liver metastases following complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal metastases: a report of three cases.World J Surg Oncol. 2019 Jun 13;17(1):99. doi: 10.1186/s12957-019-1646-0. World J Surg Oncol. 2019. PMID: 31196097 Free PMC article.
-
Status of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in patients with peritoneal carcinomatosis from colorectal cancer.J Gastrointest Oncol. 2019 Dec;10(6):1251-1265. doi: 10.21037/jgo.2019.01.36. J Gastrointest Oncol. 2019. PMID: 31949946 Free PMC article. Review.
-
Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy for Colorectal Peritoneal Metastases: A Summary of Key Clinical Trials.J Clin Med. 2022 Jun 14;11(12):3406. doi: 10.3390/jcm11123406. J Clin Med. 2022. PMID: 35743476 Free PMC article. Review.
Cited by
-
Circulating Tumor DNA as a Preoperative Marker of Recurrence in Patients with Peritoneal Metastases of Colorectal Cancer: A Clinical Feasibility Study.J Clin Med. 2020 Jun 4;9(6):1738. doi: 10.3390/jcm9061738. J Clin Med. 2020. PMID: 32512811 Free PMC article.
-
Improvement in Redox Homeostasis after Cytoreductive Surgery in Colorectal Adenocarcinoma.Oxid Med Cell Longev. 2021 Jul 31;2021:8864905. doi: 10.1155/2021/8864905. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 34381561 Free PMC article.
-
Systematic Review of Variations in Hyperthermic Intraperitoneal Chemotherapy (HIPEC) for Peritoneal Metastasis from Colorectal Cancer.J Clin Med. 2018 Dec 19;7(12):567. doi: 10.3390/jcm7120567. J Clin Med. 2018. PMID: 30572653 Free PMC article. Review.
-
Comparison of the outcomes of cytoreductive surgery versus surgery plus hyperthermic intraperitoneal chemotherapy for peritoneal carcinomatosis: a propensity score matching analysis.Surg Endosc. 2021 Jun;35(6):2789-2796. doi: 10.1007/s00464-020-07712-3. Epub 2020 Jul 6. Surg Endosc. 2021. PMID: 32632486
-
Hyperthermic intraperitoneal chemotherapy and colorectal cancer: From physiology to surgery.World J Clin Cases. 2022 Oct 26;10(30):10852-10861. doi: 10.12998/wjcc.v10.i30.10852. World J Clin Cases. 2022. PMID: 36338235 Free PMC article. Review.
References
-
- Sadeghi B, Arvieux C, Glehen O, et al. Peritoneal carcinomatosis from non-gynecologic malignancies: results of the EVOCAPE 1 multicentric prospective study. Cancer 2000;88:358-63. - PubMed
-
- American Cancer Society. Cancer Facts & Figures. 2012. Available online: http://www.cancer.org
-
- Chan CH, Cusack JC, Ryan DP. A critical look at local-regional management of peritoneal metastasis. Hematol Oncol Clin North Am 2015;29:153-8. - PubMed
-
- Esquivel J, Lowy AM, Markman M, et al. The American Society of Peritoneal Surface Malignancies (ASPSM) Multiinstitution Evaluation of the Peritoneal Surface Disease Severity Score (PSDSS) in 1,013 Patients with Colorectal Cancer with Peritoneal Carcinomatosis. Ann Surg Oncol 2014;21:4195-201. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
