Diminution of signal transducer and activator of transcription 3 signaling inhibits vascular permeability and anaphylaxis
- PMID: 26948077
- PMCID: PMC4931983
- DOI: 10.1016/j.jaci.2015.11.024
Diminution of signal transducer and activator of transcription 3 signaling inhibits vascular permeability and anaphylaxis
Erratum in
-
Correction.J Allergy Clin Immunol. 2017 Jul;140(1):320. doi: 10.1016/j.jaci.2017.05.003. J Allergy Clin Immunol. 2017. PMID: 28673402 No abstract available.
Abstract
Background: During IgE-mediated immediate hypersensitivity reactions, vascular endothelial cells permeabilize in response to mast cell mediators. We have demonstrated previously that patients and mice with signal transducer and activator of transcription 3 (STAT3) mutations (autosomal dominant hyper-IgE syndrome [AD-HIES]) are partially protected from anaphylaxis.
Objectives: We sought to study the mechanism by which STAT3 contributes to anaphylaxis and determine whether small-molecule inhibition of STAT3 can prevent anaphylaxis.
Methods: Using unaffected and STAT3-inhibited or genetic loss-of-function samples, we performed histamine skin prick tests, investigated the contribution of STAT3 to animal models of anaphylaxis, and measured endothelial cell permeability, gene and protein expression, and histamine receptor-mediated signaling.
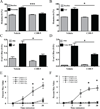
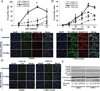
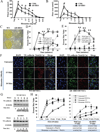
Results: Although mouse mast cell degranulation was minimally affected by STAT3 blockade, mast cell mediator-induced anaphylaxis was blunted in Stat3 mutant mice with AD-HIES and in wild-type mice subjected to small-molecule STAT3 inhibition. Histamine skin prick test responses were diminished in patients with AD-HIES. Human umbilical vein endothelial cells derived from patients with AD-HIES or treated with a STAT3 inhibitor did not signal properly through Src or cause appropriate dissolution of the adherens junctions made up of the proteins vascular endothelial-cadherin and β-catenin. Furthermore, we found that diminished STAT3 target microRNA17-92 expression in human umbilical vein endothelial cells from patients with AD-HIES is associated with increased phosphatase and tensin homolog (PTEN) expression, which inhibits Src, and increased E2F transcription factor 1 expression, which regulates β-catenin cellular dynamics.
Conclusions: These data demonstrate that STAT3-dependent transcriptional activity regulates critical components for the architecture and functional dynamics of endothelial junctions, thus permitting vascular permeability.
Keywords: Allergy; autosomal dominant hyper-IgE syndrome; immunology; innate immunity; signal transducer and activator of transcription 3.
Published by Elsevier Inc.
Figures



Similar articles
-
Diminished allergic disease in patients with STAT3 mutations reveals a role for STAT3 signaling in mast cell degranulation.J Allergy Clin Immunol. 2013 Dec;132(6):1388-96. doi: 10.1016/j.jaci.2013.08.045. Epub 2013 Nov 1. J Allergy Clin Immunol. 2013. PMID: 24184145 Free PMC article.
-
Sphingosine-1-phosphate receptor 2 protects against anaphylactic shock through suppression of endothelial nitric oxide synthase in mice.J Allergy Clin Immunol. 2013 Nov;132(5):1205-1214.e9. doi: 10.1016/j.jaci.2013.07.026. Epub 2013 Sep 8. J Allergy Clin Immunol. 2013. PMID: 24021572
-
NecroX-5 suppresses IgE/Ag-stimulated anaphylaxis and mast cell activation by regulating the SHP-1-Syk signaling module.Allergy. 2016 Feb;71(2):198-209. doi: 10.1111/all.12786. Epub 2015 Nov 23. Allergy. 2016. PMID: 26456627
-
Clinical Manifestations and Genetic Analysis of 17 Patients with Autosomal Dominant Hyper-IgE Syndrome in Mainland China: New Reports and a Literature Review.J Clin Immunol. 2017 Feb;37(2):166-179. doi: 10.1007/s10875-017-0369-7. Epub 2017 Feb 14. J Clin Immunol. 2017. PMID: 28197791 Review.
-
Deciphering the structure and function of FcεRI/mast cell axis in the regulation of allergy and anaphylaxis: a functional genomics paradigm.Cell Mol Life Sci. 2012 Jun;69(12):1917-29. doi: 10.1007/s00018-011-0886-0. Epub 2011 Dec 7. Cell Mol Life Sci. 2012. PMID: 22146792 Free PMC article. Review.
Cited by
-
Type I interferon limits mast cell-mediated anaphylaxis by controlling secretory granule homeostasis.PLoS Biol. 2019 Nov 15;17(11):e3000530. doi: 10.1371/journal.pbio.3000530. eCollection 2019 Nov. PLoS Biol. 2019. PMID: 31730616 Free PMC article.
-
Endothelial signaling by neutrophil-released oncostatin M enhances P-selectin-dependent inflammation and thrombosis.Blood Adv. 2019 Jan 22;3(2):168-183. doi: 10.1182/bloodadvances.2018026294. Blood Adv. 2019. PMID: 30670533 Free PMC article.
-
Interleukin-6 potentiates FcεRI-induced PGD2 biosynthesis and induces VEGF from human in situ-matured skin mast cells.Biochim Biophys Acta Gen Subj. 2018 May;1862(5):1069-1078. doi: 10.1016/j.bbagen.2018.01.020. Epub 2018 Feb 2. Biochim Biophys Acta Gen Subj. 2018. PMID: 29410184 Free PMC article.
-
The pathophysiology of anaphylaxis.J Allergy Clin Immunol. 2017 Aug;140(2):335-348. doi: 10.1016/j.jaci.2017.06.003. J Allergy Clin Immunol. 2017. PMID: 28780941 Free PMC article. Review.
-
Alternative Anaphylactic Routes: The Potential Role of Macrophages.Front Immunol. 2017 May 8;8:515. doi: 10.3389/fimmu.2017.00515. eCollection 2017. Front Immunol. 2017. PMID: 28533777 Free PMC article. Review.
References
-
- Takeda K, Kaisho T, Yoshida N, Takeda J, Kishimoto T, Akira S. Stat3 activation is responsible for IL-6-dependent T cell proliferation through preventing apoptosis: generation and characterization of T cell-specific Stat3-deficient mice. J Immunol. 1998;161:4652–4660. - PubMed
-
- Hirano T, Ishihara K, Hibi M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene. 2000;19:2548–2556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

