Prolyl hydroxylase domain 2 deficiency promotes skeletal muscle fiber-type transition via a calcineurin/NFATc1-dependent pathway
- PMID: 26949511
- PMCID: PMC4779261
- DOI: 10.1186/s13395-016-0079-5
Prolyl hydroxylase domain 2 deficiency promotes skeletal muscle fiber-type transition via a calcineurin/NFATc1-dependent pathway
Abstract
Background: Hypoxia exposure is known to induce an alteration in skeletal muscle fiber-type distribution mediated by hypoxia-inducible factor (HIF)-α. The downstream pathway of HIF-α leading to fiber-type shift, however, has not been elucidated. The calcineurin pathway is one of the pathways responsible for slow muscle fiber transition. Because calcineurin pathway is activated by vascular endothelial growth factor (VEGF), one of the factors induced by HIF-1α, we hypothesized that the stabilization of HIF-1α may lead to slow muscle fiber transition via the activation of calcineurin pathway in skeletal muscles. To induce HIF-1α stabilization, we used a loss of function strategy to abrogate Prolyl hydroxylase domain protein (PHD) 2 responsible for HIF-1α hydroxylation making HIF-1α susceptible to ubiquitin dependent degradation by proteasome. The purpose of this study was therefore to examine the effect of HIF-1α stabilization in PHD2 conditional knockout mouse on skeletal muscle fiber-type transition and to elucidate the involvement of calcineurin pathway on muscle fiber-type transition.
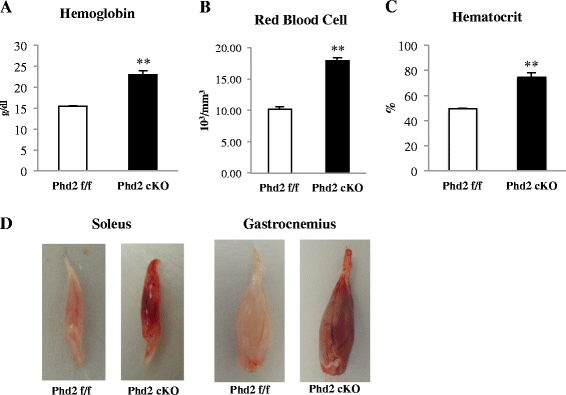
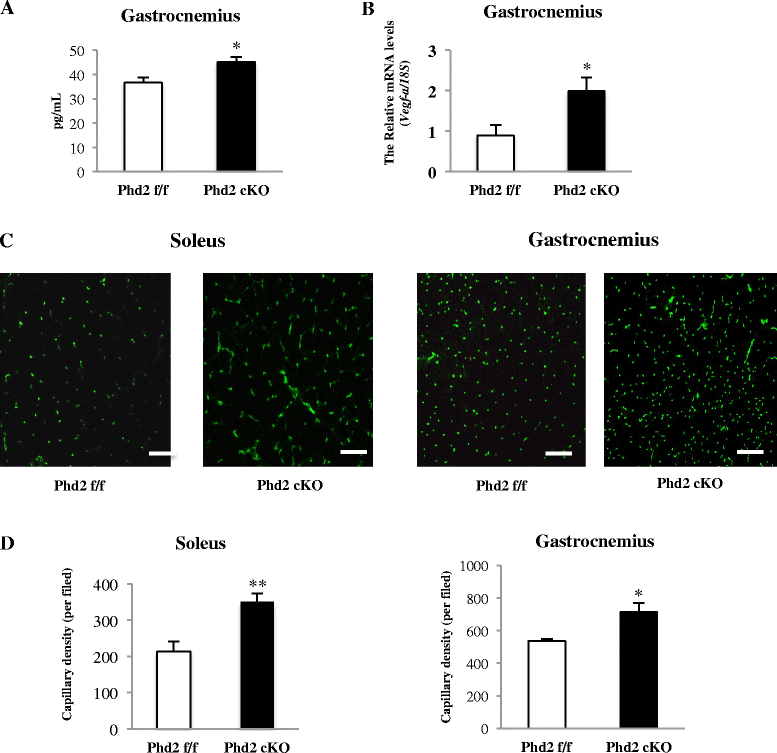
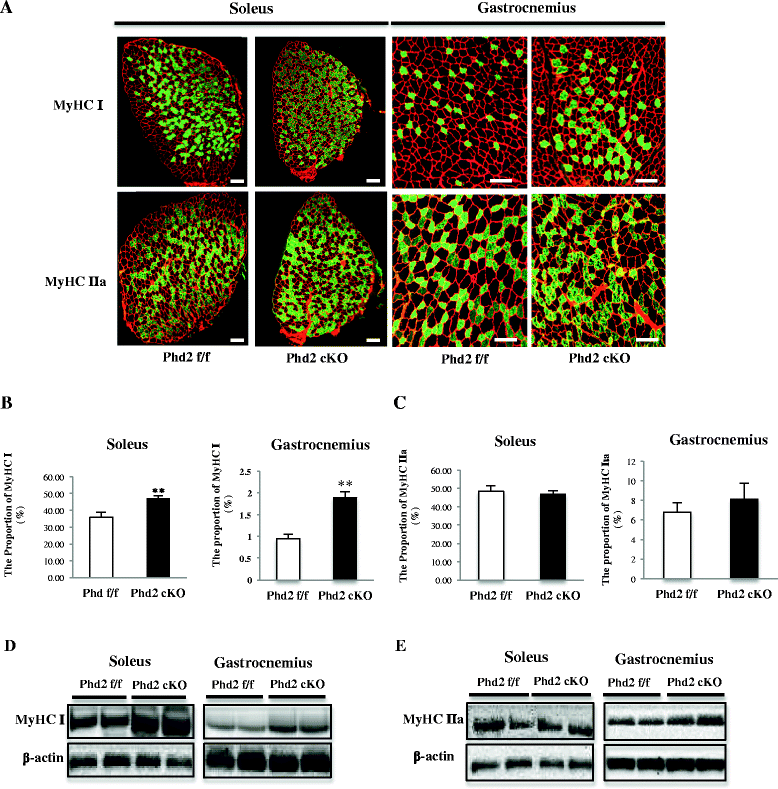
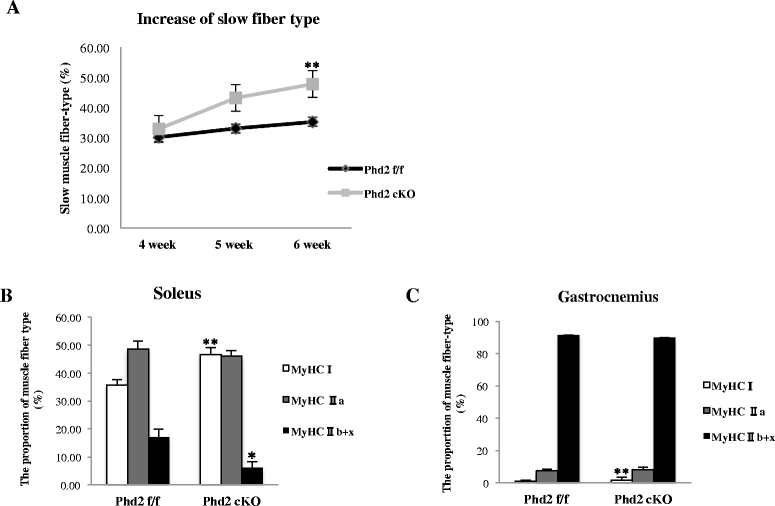
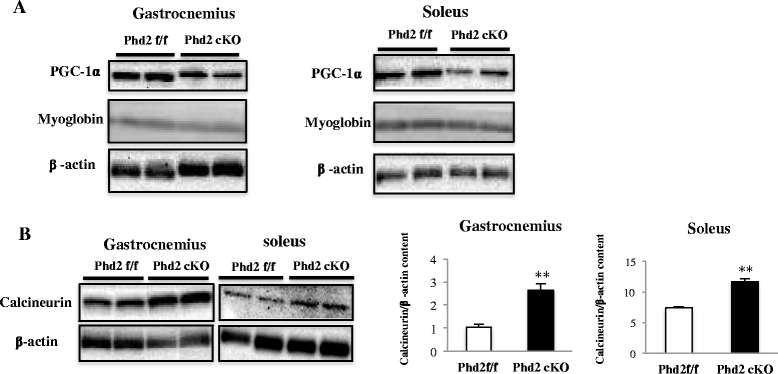
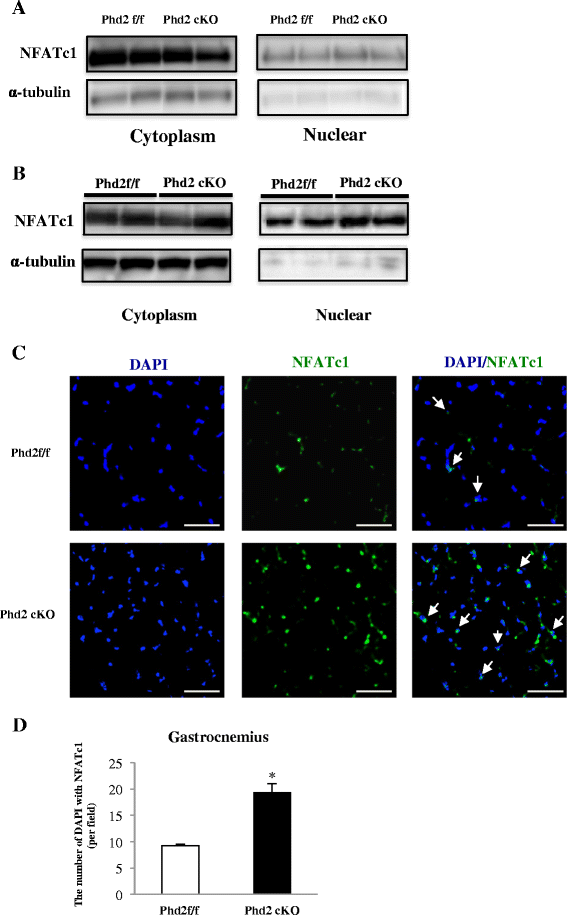
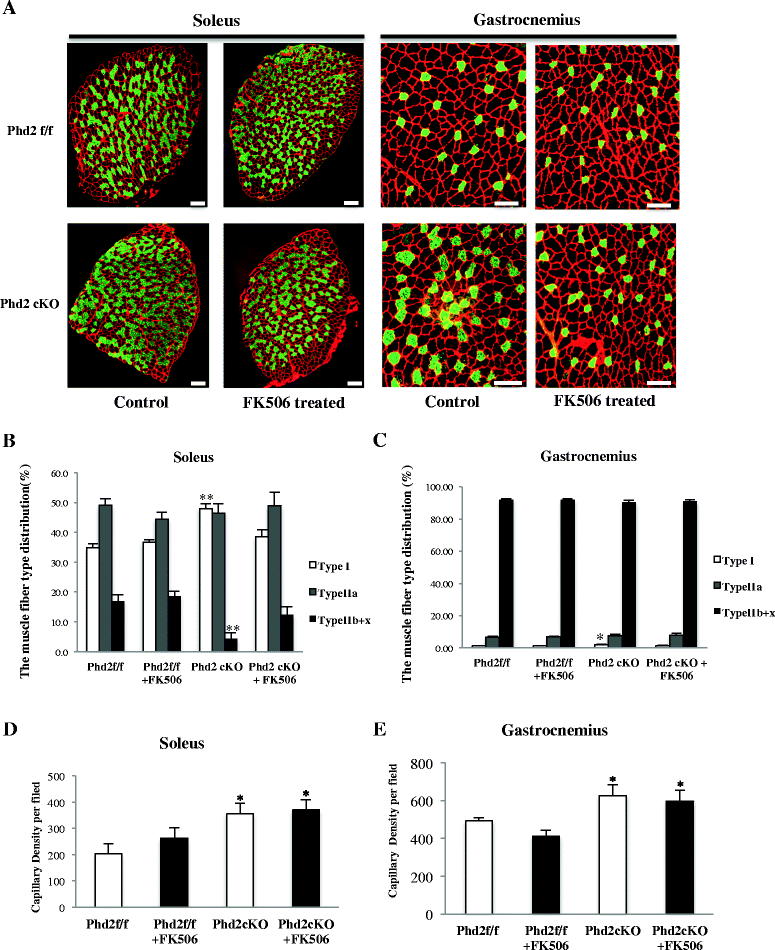
Results: PHD2 deficiency resulted in an increased capillary density in skeletal muscles due to the induction of vascular endothelial growth factor. It also elicited an alteration of skeletal muscle phenotype toward the type I fibers in both of the soleus (35.8 % in the control mice vs. 46.7 % in the PHD2-deficient mice, p < 0.01) and the gastrocnemius muscle (0.94 vs. 1.89 %, p < 0.01), and the increased proportion of type I fibers appeared to correspond to the area of increased capillary density. In addition, calcineurin and nuclear factor of activated T cell (NFATc1) protein levels were increased in both the gastrocnemius and soleus muscles, suggesting that the calcineurin/NFATc1 pathway was responsible for the type I fiber transition regardless of PGC-1α, which responded minimally to PHD2 deficiency. Indeed, we found that tacrolimus (FK-506), a calcineurin inhibitor, successfully suppressed slow fiber-type formation in PHD2-deficient mice.
Conclusions: Taken together, stabilized HIF-1α induced by PHD2 conditional knockout resulted in the transition of muscle fibers toward a slow fiber type via a calcineurin/NFATc1 signaling pathway. PHD2 conditional knockout mice may serve as a model for chronic HIF-1α stabilization as in mice exposed to low oxygen concentration.
Keywords: Calcineurin; Hypoxia-inducible factor α; NFATc1; Prolyl hydroxylate domain protein 2; Type I muscle fiber.
Figures

Similar articles
-
Specification of skeletal muscle fiber-type is determined by the calcineurin/NFATc1 signaling pathway during muscle regeneration.Biochem Biophys Res Commun. 2023 Jun 4;659:20-28. doi: 10.1016/j.bbrc.2023.03.032. Epub 2023 Mar 22. Biochem Biophys Res Commun. 2023. PMID: 37031590
-
miR-21 contributes to renal protection by targeting prolyl hydroxylase domain protein 2 in delayed ischaemic preconditioning.Nephrology (Carlton). 2017 May;22(5):366-373. doi: 10.1111/nep.12787. Nephrology (Carlton). 2017. PMID: 27030384
-
Endothelin-1 inhibits prolyl hydroxylase domain 2 to activate hypoxia-inducible factor-1alpha in melanoma cells.PLoS One. 2010 Jun 21;5(6):e11241. doi: 10.1371/journal.pone.0011241. PLoS One. 2010. Retraction in: PLoS One. 2024 Dec 5;19(12):e0315282. doi: 10.1371/journal.pone.0315282. PMID: 20574527 Free PMC article. Retracted.
-
Parallel mechanisms for resting nucleo-cytoplasmic shuttling and activity dependent translocation provide dual control of transcriptional regulators HDAC and NFAT in skeletal muscle fiber type plasticity.J Muscle Res Cell Motil. 2006;27(5-7):405-11. doi: 10.1007/s10974-006-9080-7. Epub 2006 Jul 28. J Muscle Res Cell Motil. 2006. PMID: 16874450 Review.
-
HIF-prolyl hydroxylases and cardiovascular diseases.Toxicol Mech Methods. 2012 Jun;22(5):347-58. doi: 10.3109/15376516.2012.673088. Toxicol Mech Methods. 2012. PMID: 22424133 Review.
Cited by
-
Role of brain-gut-muscle axis in human health and energy homeostasis.Front Nutr. 2022 Oct 6;9:947033. doi: 10.3389/fnut.2022.947033. eCollection 2022. Front Nutr. 2022. PMID: 36276808 Free PMC article. Review.
-
Hypoxia and Hypoxia-Inducible Factor Signaling in Muscular Dystrophies: Cause and Consequences.Int J Mol Sci. 2021 Jul 5;22(13):7220. doi: 10.3390/ijms22137220. Int J Mol Sci. 2021. PMID: 34281273 Free PMC article. Review.
-
Major Depressive Disorder and Gut Microbiota: Role of Physical Exercise.Int J Mol Sci. 2023 Nov 28;24(23):16870. doi: 10.3390/ijms242316870. Int J Mol Sci. 2023. PMID: 38069198 Free PMC article. Review.
-
Acetic acid stimulates G-protein-coupled receptor GPR43 and induces intracellular calcium influx in L6 myotube cells.PLoS One. 2020 Sep 30;15(9):e0239428. doi: 10.1371/journal.pone.0239428. eCollection 2020. PLoS One. 2020. PMID: 32997697 Free PMC article.
-
HIF-Dependent NFATC1 Activation Upregulates ITGA5 and PLAUR in Intestinal Epithelium in Inflammatory Bowel Disease.Front Genet. 2021 Nov 11;12:791640. doi: 10.3389/fgene.2021.791640. eCollection 2021. Front Genet. 2021. PMID: 34858489 Free PMC article.
References
-
- Schiaffino S, Reggiani C. Molecular diversity of myofibrillar proteins: gene regulation and functional significance. Physiol Rev. 1996;76(2):371–423. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

