β-catenin-driven binary cell fate decisions in animal development
- PMID: 26952169
- PMCID: PMC5069452
- DOI: 10.1002/wdev.228
β-catenin-driven binary cell fate decisions in animal development
Abstract
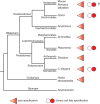
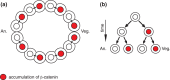
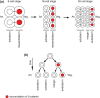
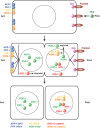
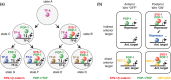
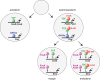
The Wnt/β-catenin pathway plays key roles during animal development. In several species, β-catenin is used in a reiterative manner to regulate cell fate diversification between daughter cells following division. This binary cell fate specification mechanism has been observed in animals that belong to very diverse phyla: the nematode Caenorhabditis elegans, the annelid Platynereis, and the ascidian Ciona. It may also play a role in the regulation of several stem cell lineages in vertebrates. While the molecular mechanism behind this binary cell fate switch is not fully understood, it appears that both secreted Wnt ligands and asymmetric cortical factors contribute to the generation of the difference in nuclear β-catenin levels between daughter cells. β-Catenin then cooperates with lineage specific transcription factors to induce the expression of novel sets of transcription factors at each round of divisions, thereby diversifying cell fate. For further resources related to this article, please visit the WIREs website.
© 2016 The Authors. WIREs Developmental Biology published by Wiley Periodicals, Inc.
Figures


References
-
- Clevers H, Nusse R. Wnt/beta‐catenin signaling and disease. Cell 2012, 149:1192–1205. - PubMed
-
- Petersen CP, Reddien PW. Wnt signaling and the polarity of the primary body axis. Cell 2009, 139:1056–1068. - PubMed
-
- Sulston JE, Schierenberg E, White JG, Thomson JN. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev Biol 1983, 100:64–119. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

