IL-19 Halts Progression of Atherosclerotic Plaque, Polarizes, and Increases Cholesterol Uptake and Efflux in Macrophages
- PMID: 26952642
- PMCID: PMC4861768
- DOI: 10.1016/j.ajpath.2015.12.023
IL-19 Halts Progression of Atherosclerotic Plaque, Polarizes, and Increases Cholesterol Uptake and Efflux in Macrophages
Abstract
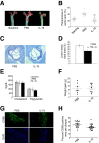
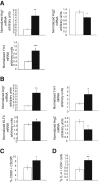
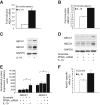
Atherosclerosis regression is an important clinical goal, and treatments that can reverse atherosclerotic plaque formation are actively being sought. Our aim was to determine whether administration of exogenous IL-19, a Th2 cytokine, could attenuate progression of preformed atherosclerotic plaque and to identify molecular mechanisms. LDLR(-/-) mice were fed a Western diet for 12 weeks, then administered rIL-19 or phosphate-buffered saline concomitant with Western diet for an additional 8 weeks. Analysis of atherosclerosis burden showed that IL-19-treated mice were similar to baseline, in contrast to control mice which showed a 54% increase in plaque, suggesting that IL-19 halted the progression of atherosclerosis. Plaque characterization showed that IL-19-treated mice had key features of atherosclerosis regression, including a reduction in macrophage content and an enrichment in markers of M2 macrophages. Mechanistic studies revealed that IL-19 promotes the activation of key pathways leading to M2 macrophage polarization, including STAT3, STAT6, Kruppel-like factor 4, and peroxisome proliferator-activated receptor γ, and can reduce cytokine-induced inflammation in vivo. We identified a novel role for IL-19 in regulating macrophage lipid metabolism through peroxisome proliferator-activated receptor γ-dependent regulation of scavenger receptor-mediated cholesterol uptake and ABCA1-mediated cholesterol efflux. These data show that IL-19 can halt progression of preformed atherosclerotic plaques by regulating both macrophage inflammation and cholesterol homeostasis and implicate IL-19 as a link between inflammation and macrophage cholesterol metabolism.
Copyright © 2016 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures




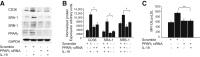
Similar articles
-
Downregulated CD36 and oxLDL uptake and stimulated ABCA1/G1 and cholesterol efflux as anti-atherosclerotic mechanisms of interleukin-10.Cardiovasc Res. 2006 Feb 1;69(2):527-35. doi: 10.1016/j.cardiores.2005.10.018. Epub 2005 Dec 5. Cardiovasc Res. 2006. PMID: 16336952
-
MicroRNA 302a is a novel modulator of cholesterol homeostasis and atherosclerosis.Arterioscler Thromb Vasc Biol. 2015 Feb;35(2):323-31. doi: 10.1161/ATVBAHA.114.304878. Epub 2014 Dec 18. Arterioscler Thromb Vasc Biol. 2015. PMID: 25524771 Free PMC article.
-
Krüppel-like factor 14 deletion in myeloid cells accelerates atherosclerotic lesion development.Cardiovasc Res. 2022 Jan 29;118(2):475-488. doi: 10.1093/cvr/cvab027. Cardiovasc Res. 2022. PMID: 33538785 Free PMC article.
-
1,25(OH)2 vitamin D suppresses macrophage migration and reverses atherogenic cholesterol metabolism in type 2 diabetic patients.J Steroid Biochem Mol Biol. 2013 Jul;136:309-12. doi: 10.1016/j.jsbmb.2012.12.019. Epub 2013 Jan 17. J Steroid Biochem Mol Biol. 2013. PMID: 23333932 Free PMC article. Review.
-
Macrophages in atherosclerosis: a dynamic balance.Nat Rev Immunol. 2013 Oct;13(10):709-21. doi: 10.1038/nri3520. Epub 2013 Sep 2. Nat Rev Immunol. 2013. PMID: 23995626 Free PMC article. Review.
Cited by
-
Interleukin-19 induces angiogenesis in the absence of hypoxia by direct and indirect immune mechanisms.Am J Physiol Cell Physiol. 2016 Jun 1;310(11):C931-41. doi: 10.1152/ajpcell.00006.2016. Epub 2016 Apr 6. Am J Physiol Cell Physiol. 2016. PMID: 27053520 Free PMC article.
-
Macrophage polarization states in atherosclerosis.Front Immunol. 2023 May 3;14:1185587. doi: 10.3389/fimmu.2023.1185587. eCollection 2023. Front Immunol. 2023. PMID: 37207214 Free PMC article. Review.
-
Treatment with IL-19 improves locomotor functional recovery after contusion trauma to the spinal cord.Br J Pharmacol. 2018 Jul;175(13):2611-2621. doi: 10.1111/bph.14193. Epub 2018 May 22. Br J Pharmacol. 2018. PMID: 29500933 Free PMC article.
-
FXR1 regulates vascular smooth muscle cell cytoskeleton, VSMC contractility, and blood pressure by multiple mechanisms.Cell Rep. 2023 Apr 25;42(4):112381. doi: 10.1016/j.celrep.2023.112381. Epub 2023 Apr 11. Cell Rep. 2023. PMID: 37043351 Free PMC article.
-
Macrophage Plasticity and Atherosclerosis Therapy.Front Mol Biosci. 2021 May 7;8:679797. doi: 10.3389/fmolb.2021.679797. eCollection 2021. Front Mol Biosci. 2021. PMID: 34026849 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

