Anti-Tumor Effects of Second Generation β-Hydroxylase Inhibitors on Cholangiocarcinoma Development and Progression
- PMID: 26954680
- PMCID: PMC4783022
- DOI: 10.1371/journal.pone.0150336
Anti-Tumor Effects of Second Generation β-Hydroxylase Inhibitors on Cholangiocarcinoma Development and Progression
Abstract
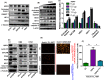
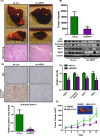
Cholangiocarcinoma (CCA) has a poor prognosis due to widespread intrahepatic spread. Aspartate β-hydroxylase (ASPH) is a transmembrane protein and catalyzes the hydroxylation of aspartyl and asparaginyl residues in calcium binding epidermal growth factor (cbEGF)-like domains of various proteins, including Notch receptors and ligands. ASPH is highly overexpressed (>95%) in human CCA tumors. We explored the molecular mechanisms by which ASPH mediated the CCA malignant phenotype and evaluated the potential of ASPH as a therapeutic target for CCA. The importance of expression and enzymatic activity of ASPH for CCA growth and progression was examined using shRNA "knockdown" and a mutant construct that reduced its catalytic activity. Second generation small molecule inhibitors (SMIs) of β-hydroxylase activity were developed and used to target ASPH in vitro and in vivo. Subcutaneous and intrahepatic xenograft rodent models were employed to determine anti-tumor effects on CCA growth and development. It was found that the enzymatic activity of ASPH was critical for mediating CCA progression, as well as inhibiting apoptosis. Mechanistically, ASPH overexpression promoted Notch activation and modulated CCA progression through a Notch1-dependent cyclin D1 pathway. Targeting ASPH with shRNAs or a SMI significantly suppressed CCA growth in vivo.
Conflict of interest statement
Figures



Similar articles
-
Aspartate β-hydroxylase targeting in castration-resistant prostate cancer modulates the NOTCH/HIF1α/GSK3β crosstalk.Carcinogenesis. 2020 Sep 24;41(9):1246-1252. doi: 10.1093/carcin/bgaa053. Carcinogenesis. 2020. PMID: 32525968
-
Aspartate beta-hydroxylase promotes cholangiocarcinoma progression by modulating RB1 phosphorylation.Cancer Lett. 2018 Aug 10;429:1-10. doi: 10.1016/j.canlet.2018.04.041. Epub 2018 May 5. Cancer Lett. 2018. PMID: 29733964 Free PMC article.
-
Targeting Aspartate Beta-Hydroxylase with the Small Molecule Inhibitor MO-I-1182 Suppresses Cholangiocarcinoma Metastasis.Dig Dis Sci. 2021 Apr;66(4):1080-1089. doi: 10.1007/s10620-020-06330-2. Epub 2020 May 22. Dig Dis Sci. 2021. PMID: 32445050
-
Diverse molecular functions of aspartate β‑hydroxylase in cancer (Review).Oncol Rep. 2020 Dec;44(6):2364-2372. doi: 10.3892/or.2020.7792. Epub 2020 Oct 6. Oncol Rep. 2020. PMID: 33125119 Free PMC article. Review.
-
Aspartate β-hydroxylase as a target for cancer therapy.J Exp Clin Cancer Res. 2020 Aug 18;39(1):163. doi: 10.1186/s13046-020-01669-w. J Exp Clin Cancer Res. 2020. PMID: 32811566 Free PMC article. Review.
Cited by
-
Aspartate β-hydroxylase Regulates Expression of Ly6 Genes.J Cancer. 2024 Jan 1;15(5):1138-1152. doi: 10.7150/jca.90422. eCollection 2024. J Cancer. 2024. PMID: 38356711 Free PMC article.
-
Notch signaling in the pathogenesis, progression and identification of potential targets for cholangiocarcinoma (Review).Mol Clin Oncol. 2022 Mar;16(3):66. doi: 10.3892/mco.2022.2499. Epub 2022 Jan 19. Mol Clin Oncol. 2022. PMID: 35154706 Free PMC article. Review.
-
Aspartate β-Hydroxylase (ASPH) Expression in Acute Myeloid Leukemia: A Potential Novel Therapeutic Target.Front Oncol. 2021 Dec 22;11:783744. doi: 10.3389/fonc.2021.783744. eCollection 2021. Front Oncol. 2021. PMID: 35004304 Free PMC article.
-
Molecular Targets and Signaling Pathways in Cholangiocarcinoma: A Systematic Review.Asian Pac J Cancer Prev. 2023 Mar 1;24(3):741-751. doi: 10.31557/APJCP.2023.24.3.741. Asian Pac J Cancer Prev. 2023. PMID: 36974526 Free PMC article.
-
Synthesis of Novel Pyridine-Carboxylates as Small-Molecule Inhibitors of Human Aspartate/Asparagine-β-Hydroxylase.ChemMedChem. 2020 Jul 3;15(13):1139-1149. doi: 10.1002/cmdc.202000147. Epub 2020 May 26. ChemMedChem. 2020. PMID: 32330361 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

