Vitronectin-Based, Biomimetic Encapsulating Hydrogel Scaffolds Support Adipogenesis of Adipose Stem Cells
- PMID: 26956095
- PMCID: PMC4841091
- DOI: 10.1089/ten.TEA.2015.0550
Vitronectin-Based, Biomimetic Encapsulating Hydrogel Scaffolds Support Adipogenesis of Adipose Stem Cells
Abstract
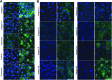
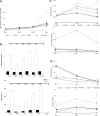
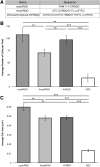
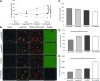
Soft tissue defects are relatively common, yet currently used reconstructive treatments have varying success rates, and serious potential complications such as unpredictable volume loss and reabsorption. Human adipose-derived stem cells (ASCs), isolated from liposuction aspirate have great potential for use in soft tissue regeneration, especially when combined with a supportive scaffold. To design scaffolds that promote differentiation of these cells down an adipogenic lineage, we characterized changes in the surrounding extracellular environment during adipogenic differentiation. We found expression changes in both extracellular matrix proteins, including increases in expression of collagen-IV and vitronectin, as well as changes in the integrin expression profile, with an increase in expression of integrins such as αVβ5 and α1β1. These integrins are known to specifically interact with vitronectin and collagen-IV, respectively, through binding to an Arg-Gly-Asp (RGD) sequence. When three different short RGD-containing peptides were incorporated into three-dimensional (3D) hydrogel cultures, it was found that an RGD-containing peptide derived from vitronectin provided strong initial attachment, maintained the desired morphology, and created optimal conditions for in vitro 3D adipogenic differentiation of ASCs. These results describe a simple, nontoxic encapsulating scaffold, capable of supporting the survival and desired differentiation of ASCs for the treatment of soft tissue defects.
Figures
Similar articles
-
Evaluation of a thermoresponsive polycaprolactone scaffold for in vitro three-dimensional stem cell differentiation.Tissue Eng Part A. 2015 Jan;21(1-2):310-9. doi: 10.1089/ten.TEA.2013.0710. Epub 2014 Nov 11. Tissue Eng Part A. 2015. PMID: 25167885
-
Hybrid Synthetic-Biological Hydrogel System for Adipose Tissue Regeneration.Macromol Biosci. 2018 Nov;18(11):e1800122. doi: 10.1002/mabi.201800122. Epub 2018 Sep 24. Macromol Biosci. 2018. PMID: 30247815 Free PMC article.
-
Strategy for constructing vascularized adipose units in poly(l-glutamic acid) hydrogel porous scaffold through inducing in-situ formation of ASCs spheroids.Acta Biomater. 2017 Mar 15;51:246-257. doi: 10.1016/j.actbio.2017.01.043. Epub 2017 Jan 16. Acta Biomater. 2017. PMID: 28093366
-
Hydrogel scaffolds for differentiation of adipose-derived stem cells.Chem Soc Rev. 2017 Oct 16;46(20):6255-6275. doi: 10.1039/c6cs00052e. Chem Soc Rev. 2017. PMID: 28816316 Review.
-
In situ Adipogenesis in Biomaterials Without Cell Seeds: Current Status and Perspectives.Front Cell Dev Biol. 2021 Mar 8;9:647149. doi: 10.3389/fcell.2021.647149. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33763426 Free PMC article. Review.
Cited by
-
Cell-mediated remodeling of biomimetic encapsulating hydrogels triggered by adipogenic differentiation of adipose stem cells.J Tissue Eng. 2016 Sep 23;7:2041731416670482. doi: 10.1177/2041731416670482. eCollection 2016 Jan-Dec. J Tissue Eng. 2016. PMID: 27733898 Free PMC article.
-
Human Adipose-Derived Stromal/Stem Cell Culture and Analysis Methods for Adipose Tissue Modeling In Vitro: A Systematic Review.Cells. 2021 Jun 3;10(6):1378. doi: 10.3390/cells10061378. Cells. 2021. PMID: 34204869 Free PMC article.
-
Unleashing the potential of adipose organoids: A revolutionary approach to combat obesity-related metabolic diseases.Theranostics. 2024 Feb 25;14(5):2075-2098. doi: 10.7150/thno.93919. eCollection 2024. Theranostics. 2024. PMID: 38505622 Free PMC article. Review.
-
Effects of tunable, 3D-bioprinted hydrogels on human brown adipocyte behavior and metabolic function.Acta Biomater. 2018 Apr 15;71:486-495. doi: 10.1016/j.actbio.2018.03.021. Epub 2018 Mar 16. Acta Biomater. 2018. PMID: 29555462 Free PMC article.
-
Synthetic fibrous hydrogels as a platform to decipher cell-matrix mechanical interactions.Proc Natl Acad Sci U S A. 2023 Apr 11;120(15):e2216934120. doi: 10.1073/pnas.2216934120. Epub 2023 Apr 3. Proc Natl Acad Sci U S A. 2023. PMID: 37011188 Free PMC article.
References
-
- Yoshimura K., Asano Y., Aoi N., Kurita M., Oshima Y., Sato K., et al. . Progenitor-enriched adipose tissue transplantation as rescue for breast implant complications. Breast J 16, 169, 2010 - PubMed
-
- Oedayrajsingh-Varma M.J., van Ham S.M., Knippenberg M., Helder M.N., Klein-Nulend J., Schouten T.E., et al. . Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure. Cytotherapy 8, 166, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

