Mechanical oscillations enhance gene delivery into suspended cells
- PMID: 26956215
- PMCID: PMC4783776
- DOI: 10.1038/srep22824
Mechanical oscillations enhance gene delivery into suspended cells
Abstract
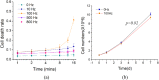
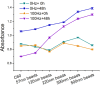
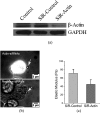
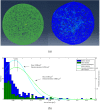
Suspended cells are difficult to be transfected by common biochemical methods which require cell attachment to a substrate. Mechanical oscillations of suspended cells at certain frequencies are found to result in significant increase in membrane permeability and potency for delivery of nano-particles and genetic materials into the cells. Nanomaterials including siRNAs are found to penetrate into suspended cells after subjecting to short-time mechanical oscillations, which would otherwise not affect the viability of the cells. Theoretical analysis indicates significant deformation of the actin-filament network in the cytoskeleton cortex during mechanical oscillations at the experimental frequency, which is likely to rupture the soft phospholipid bilayer leading to increased membrane permeability. The results here indicate a new method for enhancing cell transfection.
Figures


References
-
- Putnam D. Polymers for gene delivery across length scales. Nat Mater 5, 439–451 (2006). - PubMed
-
- Jiang M. & Chen G. High Ca2+-phosphate transfection efficiency in low-density neuronal cultures. Nat Protoc 1, 695–700 (2006). - PubMed
-
- Rahim A. A. et al. Efficient gene delivery to the adult and fetal CNS using pseudotyped non-integrating lentiviral vectors. Gene Ther 16, 509–520 (2009). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

