Oral Administration of p-Hydroxycinnamic Acid Attenuates Atopic Dermatitis by Downregulating Th1 and Th2 Cytokine Production and Keratinocyte Activation
- PMID: 26959360
- PMCID: PMC4784746
- DOI: 10.1371/journal.pone.0150952
Oral Administration of p-Hydroxycinnamic Acid Attenuates Atopic Dermatitis by Downregulating Th1 and Th2 Cytokine Production and Keratinocyte Activation
Abstract
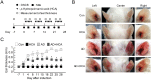
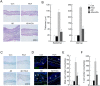
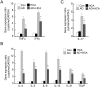
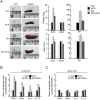
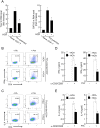
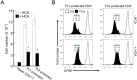
Atopic dermatitis (AD) is a complex disease that is caused by various factors, including environmental change, genetic defects, and immune imbalance. We previously showed that p-hydroxycinnamic acid (HCA) isolated from the roots of Curcuma longa inhibits T-cell activation without inducing cell death. Here, we demonstrated that oral administration of HCA in a mouse model of ear AD attenuates the following local and systemic AD manifestations: ear thickening, immune-cell infiltration, production of AD-promoting immunoregulatory cytokines in ear tissues, increased spleen and draining lymph node size and weight, increased pro-inflammatory cytokine production by draining lymph nodes, and elevated serum immunoglobulin production. HCA treatment of CD4+ T cells in vitro suppressed their proliferation and differentiation into Th1 or Th2 and their Th1 and Th2 cytokine production. HCA treatment of keratinocytes lowered their production of the pro-inflammatory cytokines that drive either Th1 or Th2 responses in AD. Thus, HCA may be of therapeutic potential for AD as it acts by suppressing keratinocyte activation and downregulating T-cell differentiation and cytokine production.
Conflict of interest statement
Figures
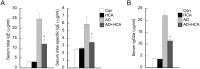

Similar articles
-
Oral Administration of 4-Hydroxy-3-Methoxycinnamaldehyde Attenuates Atopic Dermatitis by Inhibiting T Cell and Keratinocyte Activation.PLoS One. 2015 Dec 10;10(12):e0144521. doi: 10.1371/journal.pone.0144521. eCollection 2015. PLoS One. 2015. PMID: 26656486 Free PMC article.
-
Chrysin attenuates atopic dermatitis by suppressing inflammation of keratinocytes.Food Chem Toxicol. 2017 Dec;110:142-150. doi: 10.1016/j.fct.2017.10.025. Epub 2017 Oct 16. Food Chem Toxicol. 2017. PMID: 29050978
-
Induction of GITRL expression in human keratinocytes by Th2 cytokines and TNF-α: implications for atopic dermatitis.Clin Exp Allergy. 2012 Apr;42(4):550-9. doi: 10.1111/j.1365-2222.2012.03956.x. Clin Exp Allergy. 2012. PMID: 22417213 Free PMC article.
-
The role of cytokines/chemokines in the pathogenesis of atopic dermatitis.Curr Probl Dermatol. 2011;41:80-92. doi: 10.1159/000323299. Epub 2011 May 12. Curr Probl Dermatol. 2011. PMID: 21576949 Review.
-
[Th1 cells, Th2 cells and atopic dermatitis].Hautarzt. 1997 Apr;48(4):223-7. doi: 10.1007/s001050050573. Hautarzt. 1997. PMID: 9206708 Review. German.
Cited by
-
Chrysophanol Attenuates Manifestations of Immune Bowel Diseases by Regulation of Colorectal Cells and T Cells Activation In Vivo.Molecules. 2021 Mar 17;26(6):1682. doi: 10.3390/molecules26061682. Molecules. 2021. PMID: 33802855 Free PMC article.
-
Integrated plasma metabolomic and cytokine analysis reveals a distinct immunometabolic signature in atopic dermatitis.Front Immunol. 2024 Mar 15;15:1354128. doi: 10.3389/fimmu.2024.1354128. eCollection 2024. Front Immunol. 2024. PMID: 38558806 Free PMC article.
-
Pterostilbene reduces the progression of atopic dermatitis via modulating inflammatory and oxidative stress biomarkers in mice.Inflammopharmacology. 2023 Jun;31(3):1289-1303. doi: 10.1007/s10787-023-01214-z. Epub 2023 Apr 17. Inflammopharmacology. 2023. PMID: 37069463
-
Investigating efficacy of "microbiota modulation of the gut-lung Axis" combined with chemotherapy in patients with advanced NSCLC: study protocol for a multicenter, prospective, double blind, placebo controlled, randomized trial.BMC Cancer. 2021 Jun 22;21(1):721. doi: 10.1186/s12885-021-08448-6. BMC Cancer. 2021. PMID: 34157996 Free PMC article. Clinical Trial.
-
Galectin-9 Induced by Dietary Probiotic Mixture Regulates Immune Balance to Reduce Atopic Dermatitis Symptoms in Mice.Front Immunol. 2020 Jan 22;10:3063. doi: 10.3389/fimmu.2019.03063. eCollection 2019. Front Immunol. 2020. PMID: 32038618 Free PMC article.
References
-
- Matsumoto M, Ra C, Kawamoto K, Sato H, Itakura A, Sawada J, et al. IgE hyperproduction through enhanced tyrosine phosphorylation of Janus kinase 3 in NC/Nga mice, a model for human atopic dermatitis. J Immunol. 1999;162: 1056–1063. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

