Pharmacological treatment for Buerger's disease
- PMID: 26967103
- PMCID: PMC7104255
- DOI: 10.1002/14651858.CD011033.pub3
Pharmacological treatment for Buerger's disease
Update in
-
Pharmacological treatment for Buerger's disease.Cochrane Database Syst Rev. 2020 May 4;5(5):CD011033. doi: 10.1002/14651858.CD011033.pub4. Cochrane Database Syst Rev. 2020. PMID: 32364620 Free PMC article.
Abstract
Background: Buerger's disease (thromboangiitis obliterans) is a non-atherosclerotic, segmental inflammatory pathology that most commonly affects the small and medium sized arteries, veins, and nerves in the upper and lower extremities. The etiology is unknown, but involves hereditary susceptibility, tobacco exposure, immune and coagulation responses. In many cases, there is no possibility of revascularization to improve the condition. Pharmacological treatment is an option for patients with severe complications, such as ischaemic ulcers or rest pain.
Objectives: To assess the effectiveness of any pharmacological agent (intravenous or oral) compared with placebo or any other pharmacological agent in patients with Buerger's disease.
Search methods: The Cochrane Vascular Trials Search Co-ordinator searched their Specialised Register (last searched in April 2015) and the Cochrane Register of Studies (Issue 3, 2015). The review authors searched trial registers and the European grey literature; screened reference lists of relevant studies, and contacted study authors and major pharmaceutical companies.
Selection criteria: Randomised controlled trials (RCTs) involving pharmacological agents used in the treatment of Buerger's disease.
Data collection and analysis: Two review authors, independently assessed the studies, extracted data and performed data analysis.
Main results: Five randomised controlled trials (total 602 participants) compared prostacyclin analogue with placebo, aspirin, or a prostaglandin analogue, and folic acid with placebo. No studies assessed other pharmacological agents such as cilostazol, clopidogrel and pentoxifylline or compared oral versus intravenous prostanoid.Compared with aspirin, intravenous prostacyclin analogue iloprost improved ulcer healing (risk ratio (RR) 2.65; 95% confidence interval (CI) 1.15 to 6.11; 98 participants; one study; moderate quality evidence), and helped to eradicate rest pain after 28 days (RR 2.28; 95% CI 1.48 to 3.52; 133 participants; one study; moderate quality evidence), although amputation rates were similar six months after treatment (RR 0.32; 95% CI 0.09 to 1.15; 95 participants; one study; moderate quality evidence). When comparing prostacyclin (iloprost and clinprost) with prostaglandin (alprostadil) analogues, ulcer healing was similar (RR 1.13; 95% CI 0.76 to 1.69; 89 participants; two studies; I² = 0%; very low quality evidence), as was the eradication of rest pain after 28 days (RR 1.57; 95% CI 0.72 to 3.44; 38 participants; one study; low quality evidence), while amputation rates were not measured. Compared with placebo, the effects of oral prostacyclin analogue iloprost were similar for: healing ischaemic ulcers (iloprost 200 mcg: RR 1.11; 95% CI 0.54 to 2.29; 133 participants; one study; moderate quality evidence, and iloprost 400 mcg: RR 0.90; 95% CI 0.42 to 1.93; 135 participants; one study; moderate quality evidence), eradication of rest pain after eight weeks (iloprost 200 mcg: RR 1.14; 95% CI 0.79 to 1.63; 207 participants; one study; moderate quality evidence, and iloprost 400 mcg: RR 1.11; 95% CI 0.77 to 1.59; 201 participants; one study; moderate quality evidence), and amputation rates after six months (iloprost 200 mcg: RR 0.54; 95% CI 0.19 to 1.56; 209 participants; one study, and iloprost 400 mcg: RR 0.42; 95% CI 0.13 to 1.31; 213 participants; one study). When comparing folic acid with placebo in patients with Buerger's disease and hyperhomocysteinaemia, pain scores were similar, there were no new cases of amputation in either group, and ulcer healing was not assessed (very low quality evidence).Treatment side effects such as headaches, flushing or nausea were not associated with treatment interruptions or more serious consequences. Outcomes such as amputation-free survival, walking distance or pain-free walking distance, and ankle brachial index were not assessed by any study.Overall, the quality of the evidence was very low to moderate, with few studies, small numbers of participants, variation in severity of disease of participants between studies and missing information regarding for example baseline tobacco exposure.
Authors' conclusions: Moderate quality evidence suggests that intravenous iloprost (prostacyclin analogue) is more effective than aspirin for eradicating rest pain and healing ischaemic ulcers in Buerger's disease, but oral iloprost is not more effective than placebo. Verylow and low quality evidence suggests there is no difference between prostacyclin (iloprost and clinprost) and the prostaglandin analogue alprostadil for healing ulcers and relieving pain respectively in severe Buerger's disease. Very-low quality evidence suggests there is no difference in pain scores and amputation rates between folic acid and placebo, in people with Buerger's disease and hyperhomocysteinaemia. High quality trials assessing the effectiveness of pharmacological agents (intravenous or oral) in people with Buerger's disease are needed.
Conflict of interest statement
DGC: none known CRM: none known JCCBS: none known
Figures



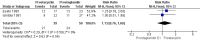


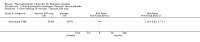





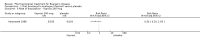




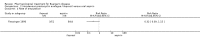







Update of
-
Pharmacological treatment for Buerger's disease.Cochrane Database Syst Rev. 2016 Feb 1;2:CD011033. doi: 10.1002/14651858.CD011033.pub2. Cochrane Database Syst Rev. 2016. Update in: Cochrane Database Syst Rev. 2016 Mar 11;3:CD011033. doi: 10.1002/14651858.CD011033.pub3. PMID: 26828199 Updated.
Similar articles
-
Pharmacological treatment for Buerger's disease.Cochrane Database Syst Rev. 2016 Feb 1;2:CD011033. doi: 10.1002/14651858.CD011033.pub2. Cochrane Database Syst Rev. 2016. Update in: Cochrane Database Syst Rev. 2016 Mar 11;3:CD011033. doi: 10.1002/14651858.CD011033.pub3. PMID: 26828199 Updated.
-
Pharmacological treatment for Buerger's disease.Cochrane Database Syst Rev. 2020 May 4;5(5):CD011033. doi: 10.1002/14651858.CD011033.pub4. Cochrane Database Syst Rev. 2020. PMID: 32364620 Free PMC article.
-
Lumbar sympathectomy versus prostanoids for critical limb ischaemia due to non-reconstructable peripheral arterial disease.Cochrane Database Syst Rev. 2018 Apr 16;4(4):CD009366. doi: 10.1002/14651858.CD009366.pub2. Cochrane Database Syst Rev. 2018. PMID: 29658630 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
Cited by
-
The Buerger's rabbit model: a closer step to unravelling thromboangiitis obliterans?Thromb J. 2024 Jul 29;22(1):69. doi: 10.1186/s12959-024-00638-z. Thromb J. 2024. PMID: 39075498 Free PMC article.
-
Assessing Mailuoning injection in wound healing and thrombophlebitis management: A rat model study.Int Wound J. 2024 Apr;21(4):e14527. doi: 10.1111/iwj.14527. Epub 2023 Dec 14. Int Wound J. 2024. Retraction in: Int Wound J. 2025 Apr;22(4):e70476. doi: 10.1111/iwj.70476. PMID: 38095110 Free PMC article. Retracted.
-
Surgical Treatment Options for Buerger's Disease (Experience with 315 Cases in Iran).Med J Islam Repub Iran. 2022 Nov 14;36:134. doi: 10.47176/mjiri.36.134. eCollection 2022. Med J Islam Repub Iran. 2022. PMID: 36479532 Free PMC article.
-
Efficacy of bosentan in patients with refractory thromboangiitis obliterans (Buerger disease): A case series and review of the literature.Medicine (Baltimore). 2016 Nov;95(48):e5511. doi: 10.1097/MD.0000000000005511. Medicine (Baltimore). 2016. PMID: 27902617 Free PMC article. Review.
-
Marburg and Ebola Virus Infections Elicit a Complex, Muted Inflammatory State in Bats.Viruses. 2023 Jan 26;15(2):350. doi: 10.3390/v15020350. Viruses. 2023. PMID: 36851566 Free PMC article.
References
References to studies included in this review
Beigi 2014 {published data only}
Esato 1995 {published data only}
-
- Esato K, Yasuda K, Abe T, Hoshino S, Ishimaru S, Hori G, et al. Studies on the clinical efficacy and safety of TTC‐909 in the treatment of peripheral arterial occlusive disease: a multi‐center double‐blind comparison with Alprostadil incorporated in lipid microspheres. Rinsho Iyaku 1995;11:2111‐2.
Fiessinger 1990 {published data only}
-
- Diehm C. Multicenter‐study on Iloprost in patients with thromboangiitis obliterans (Buerger's disease). Klinische Wochenschrift 1989;67:178‐9.
-
- Fiessinger JN, Schafer M. Trial of iloprost versus aspirin treatment for critical limb ischaemia of thromboangiitis obliterans. Lancet 1990;335(8689):555‐7. - PubMed
Ishitobi 1991 {published data only}
-
- Ishitobi K, et al. Therapeutic efficacy of iloprost, a prostacyclin analogue for chronic arterial occlusion: a double‐blind comparative study with PGE1. Rinsho to Kenkyu 1991;68:1836‐50.
Verstraete 1998 {published data only}
-
- Verstraete M. Oral iloprost in the treatment of thromboangiitis obliterans (Buerger's disease): A double‐blind, randomised, placebo‐controlled trial. The European TAO Study Group. European Journal of Vascular and Endovascular Surgery 1998;15(4):300‐7. - PubMed
References to studies excluded from this review
Bozkurt 2006 {published data only}
-
- Bozkurt AK, Koksal C, Demirbas MY, Erdogan A, Rahman A, Demirkilic U, et al. A randomized trial of intravenous iloprost (a stable prostacyclin analogue) versus lumbar sympathectomy in the management of Buerger's disease. International Angiology 2006;25:162‐8. - PubMed
Coscia 1972 {published data only}
-
- Coscia M. Effects of nicergoline in peripheral arteriopathies of the lower limbs. Double‐blind study. Minerva Cardioangiologica 1972;20(9):497‐504. - PubMed
He 2007 {published data only}
-
- He D. The application of Simiaoyongantangjiawei in the treatment of thromboangiitis obliterans. Chinese Journal of Information on Traditional Chinese Medicine 2007;14:76.
Musial 1986 {published data only}
-
- Musial J, Wilczynska M, Sladek K, Cierniewski CS, Nizankowski R, Szczeklik A. Fibrinolytic activity of prostacyclin and iloprost in patients with peripheral arterial disease. Prostaglandins 1986;31(1):61‐70. - PubMed
Reichert 1975 {published data only}
-
- Reichert N, Shibolet S, Adar R, Gafni J. Controlled trial of propranolol in intermittent claudication. Clinical Pharmacology and Therapeutics 1975;17:612‐5. - PubMed
Steinorth 1967 {published data only}
-
- Steinorth G. Report on the clinical testing of Lamuran (I). Die Medizinische Welt 1967;35:2025‐32. - PubMed
Sun 1993 {published data only}
-
- Sun DX, Mu DJ, Li XY. Point injection of prostaglandin E1 on Sanyinjiao Acupuncture (SP6) for thromboangiitis obliterans pain in 24 cases. Zhongguo Zhong Xi Yi Jie He Za Zhi [Chinese Journal of Integrated Traditional and Western Medicine] 1993;13(10).
Yang 2005 {published data only}
-
- Yang L, Zhou CH, Yao T. Clinical study of treating thromboangitis obliterans with overlapping thinning chisel therapy. Chinese Journal of Information on TCM 2005;12:73‐4.
Zelikovsky 1973 {published data only}
-
- Zelikovsky A, Urca I, Shaklai M, Vries A. A clinical trial with pyridinolcarbamate in the treatment of patients with obstructive diseases of the peripheral arteries. Japanese Heart Journal 1973;14:12‐21. - PubMed
References to studies awaiting assessment
Hoshino 1997 {published data only}
-
- Hoshino S, Nakashima M, Kubo Y, Yasuda K, Abe T, Ouchi H, et al. Results of dose finding study of Pamicogrel (KBT‐3022) on chronic arterial occlusive diseases. Rinsho Iyaku 1997;13:3409‐36.
Additional references
Atkins 2004
Bartolo 1987
-
- Bartolo M, Antignani PL, Todini AR, Ricci G. Buerger's disease: etiologic role of the rickettsiae?. Journal des Maladies Vasculaires 1987;12(1):82‐4. - PubMed
Bedenis 2014
Brunton 2011
-
- Brunton LL, Chabner BA, Knollmann BC (editors). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 12th Edition. New York: McGraw‐Hill Companies, 2011.
Buerger 1908
-
- Buerger L. Thrombo‐angiitis obliterans: a study of the vascular lesions leading to presenile spontaneous gangrene. American Journal of Medicine 1908;136:567‐80. - PubMed
Cachovan 1988
-
- Cachovan M. Epidemiology and geographic distribution of the thromboangiitis obliterans [Epidemiologic und geographisches Verteilungsmuster der thromboangiitis obliterans]. Thromboangiitis obliterans Morbus Winiwarter‐Buerger. Heidrich H. Stuttgart, 1988:31‐6.
Chapman 2003
-
- Chapman TM, Goa KL. Cilostazol: a review of its use in intermittent claudication. American Journal of Cardiovascular Drugs 2003;3(2):117‐38. - PubMed
Cooper 2004
-
- Cooper LT, Tse TS, Mikhail MA, McBane RD, Stanson AW, Ballman KV. Long‐term survival and amputation risk in thromboangiitis obliterans (Buerger's disease). Journal of the American College of Cardiology 2004;44(12):2410‐1. - PubMed
De Haro 2009
Dindyal 2009
-
- Dindyal S, Kyriakides C. A review of cilostazol, a phosphodiesterase inhibitor, and its role in preventing both coronary and peripheral arterial restenosis following endovascular therapy. Recent Patents on Cardiovascular Drug Discovery 2009;4(1):6‐14. - PubMed
Fazeli 2013
-
- Fazeli B. Is rickettsia the key to solving the puzzle of Buerger’s disease?. Vascular 2013;22(5):393‐4. - PubMed
FDA 1999
-
- US Food, Drug Administration. FDA approved drug products. http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseacti.... DrugDetails (accessed 25 November 2015).
Fontaine 1954
-
- Fontaine R, Kim M, Kieny R. Surgical treatment of peripheral circulation disorders [Die chirugische Behandlung der peripheren Durchblutungsstörungen]. Helvetica Chirurgica Acta 1954;21(5/6):499‐533. - PubMed
Grant 1992
-
- Grant SM, Goa KL. Iloprost. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in peripheral vascular disease, myocardial ischaemia and extracorporeal circulation procedures. Drugs 1992;43(6):889‐924. - PubMed
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Iwai 2005
-
- Iwai T, Inoue Y, Umeda M, Huang Y, Kurihara N, Koike M, et al. Oral bacteria in the occluded arteries of patients with Buerger disease. Journal of Vascular Surgery 2005;42(1):107‐15. - PubMed
Launay 2006
-
- Launay D, Diot E, Pasquier E, Mouthon L, Boullanger N, Fain O, et al. Bosentan for treatment of active digital ulcers in patients with systemic sclerosis. La Presse Médicale 2006;35(4 Pt 1):587‐92. - PubMed
Li 2008
-
- Li X, Iwai T, Nakamura H, Inoue Y, Chen Y, Umeda M, et al. An ultrastructural study of Porphyromonas gingivalis‐induced platelet aggregation. Thrombosis Research 2008;122(6):810‐9. - PubMed
Malecki 2009
-
- Malecki R, Zdrojowy K, Adamiec R. Thromboangiitis obliterans in the 21st century ‐ a new face of disease. Atherosclerosis 2009;206(2):328‐34. - PubMed
Matucci‐Cerinic 2011
Novo 2004
-
- Novo S, Coppola G, Milio G. Critical limb ischemia: definition and natural history. Current Drug Targets 2004;4(3):219‐25. - PubMed
Olin 2000
-
- Olin JW. Thromboangiitis obliterans (Buerger's disease). New England Journal of Medicine 2000;343(12):864‐9. - PubMed
Olin 2006
-
- Olin JW, Shih A. Thromboangiitis obliterans (Buerger’s disease). Current Opinion in Rheumatology 2006;18(1):18‐24. - PubMed
Ruffolo 2010
Rutherford 2005
-
- Rutherford RB. Vascular Surgery. 6th Edition. Vol. 1, New York: Elsevier Inc, 2005.
Safdar 2011
-
- Safdar Z. Treatment of pulmonary arterial hypertension: the role of prostacyclin and prostaglandin analogs. Respiratory Medicine 2011;105(6):818‐27. - PubMed
Shionoya 1983
-
- Shionoya S. What is Buerger's disease?. World Journal of Surgery 1983;7(4):544‐51. - PubMed
von Winiwarter 1879
-
- Winiwarter F. A peculiar form of endarteritis and endophlebitis with gangrene of the foot [Ueber eine eigenthümliche Form von Endarteriitis und Endophlebitis mit Gangrän des Fusses]. Archiv für Klinische Chirurgie 1879;23:202‐26.
Weber 1996
-
- Weber C, Schmitt R, Birnboeck H, Hopfgartner G, Marle SP, Peeters PAM, et al. Pharmacokinetics and pharmacodynamics of the endothelin‐receptor antagonist bosentanin healthy human subjects. Clinical Pharmacology and Therapeutics 1996;60:124‐37. - PubMed
Weinberg 2012
-
- Weinberg I, Jaff MR. Nonatherosclerotic arterial disorders of the lower extremities. Circulation 2012;126(2):213‐22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

