A systematic review of the characteristics and validity of monitoring technologies to assess Parkinson's disease
- PMID: 26969628
- PMCID: PMC4788909
- DOI: 10.1186/s12984-016-0136-7
A systematic review of the characteristics and validity of monitoring technologies to assess Parkinson's disease
Erratum in
-
Erratum to: A systematic review of the characteristics and validity of monitoring technologies to assess Parkinson's disease.J Neuroeng Rehabil. 2016 Aug 1;13(1):71. doi: 10.1186/s12984-016-0181-2. J Neuroeng Rehabil. 2016. PMID: 27480970 Free PMC article. No abstract available.
Abstract
Background: There is growing interest in having objective assessment of health-related outcomes using technology-based devices that provide unbiased measurements which can be used in clinical practice and scientific research. Many studies have investigated the clinical manifestations of Parkinson's disease using such devices. However, clinimetric properties and clinical validation vary among the different devices.
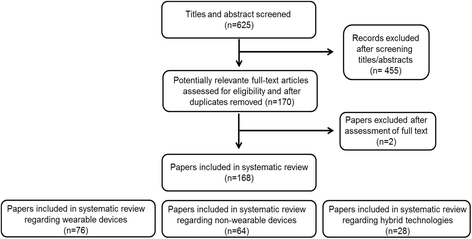
Methods: Given such heterogeneity, we sought to perform a systematic review in order to (i) list, (ii) compare and (iii) classify technological-based devices used to measure motor function in individuals with Parkinson's disease into three groups, namely wearable, non-wearable and hybrid devices. A systematic literature search of the PubMed database resulted in the inclusion of 168 studies. These studies were grouped based on the type of device used. For each device we reviewed availability, use, reliability, validity, and sensitivity to change. The devices were then classified as (i) 'recommended', (ii) 'suggested' or (iii) 'listed' based on the following criteria: (1) used in the assessment of Parkinson's disease (yes/no), (2) used in published studies by people other than the developers (yes/no), and (3) successful clinimetric testing (yes/no).
Results: Seventy-three devices were identified, 22 were wearable, 38 were non-wearable, and 13 were hybrid devices. In accordance with our classification method, 9 devices were 'recommended', 34 devices were 'suggested', and 30 devices were classified as 'listed'. Within the wearable devices group, the Mobility Lab sensors from Ambulatory Parkinson's Disease Monitoring (APDM), Physilog®, StepWatch 3, TriTrac RT3 Triaxial accelerometer, McRoberts DynaPort, and Axivity (AX3) were classified as 'recommended'. Within the non-wearable devices group, the Nintendo Wii Balance Board and GAITRite® gait analysis system were classified as 'recommended'. Within the hybrid devices group only the Kinesia® system was classified as 'recommended'.
Keywords: Monitoring technologies; Outcomes; Parkinson’s disease; Quantitative assessment; Wearable devices.
References
-
- Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23(15):2129–70. doi: 10.1002/mds.22340. - DOI - PubMed
-
- Mokkink LB, Terwee CB, Patrick DL, Alonso J, Stratford PW, Knol DL, et al. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J Clin Epidemiol. 2010;63:737–45. doi: 10.1016/j.jclinepi.2010.02.006. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous