Gender Dependent Evaluation of Autism like Behavior in Mice Exposed to Prenatal Zinc Deficiency
- PMID: 26973485
- PMCID: PMC4776245
- DOI: 10.3389/fnbeh.2016.00037
Gender Dependent Evaluation of Autism like Behavior in Mice Exposed to Prenatal Zinc Deficiency
Abstract
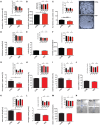
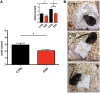
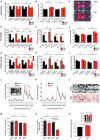
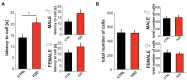
Zinc deficiency has recently been linked to the etiology of autism spectrum disorders (ASD) as environmental risk factor. With an estimated 17% of the world population being at risk of zinc deficiency, especially zinc deficiency during pregnancy might be a common occurrence, also in industrialized nations. On molecular level, zinc deficiency has been shown to affect a signaling pathway at glutamatergic synapses that has previously been identified through genetic mutations in ASD patients, the Neurexin-Neuroligin-Shank pathway, via altering zinc binding Shank family members. In particular, prenatal zinc deficient but not acute zinc deficient animals have been reported to display autism like behavior in some behavioral tests. However, a full behavioral analysis of a possible autism like behavior has been lacking so far. Here, we performed an extensive behavioral phenotyping of mice born from mothers with mild zinc deficiency during all trimesters of pregnancy. Prenatal zinc deficient animals were investigated as adults and gender differences were assessed. Our results show that prenatal zinc deficient mice display increased anxiety, deficits in nest building and various social interaction paradigm, as well as mild alterations in ultrasonic vocalizations. A gender specific analysis revealed only few sex specific differences. Taken together, given that similar behavioral abnormalities as reported here are frequently observed in ASD mouse models, we conclude that prenatal zinc deficient animals even without specific genetic susceptibility for ASD, already show some features of ASD like behavior.
Keywords: ASD; ZIP; Zn; Zn2+; ZnT; brain; plasticity.
Figures


Similar articles
-
Zinc deficiency dysregulates the synaptic ProSAP/Shank scaffold and might contribute to autism spectrum disorders.Brain. 2014 Jan;137(Pt 1):137-52. doi: 10.1093/brain/awt303. Epub 2013 Nov 25. Brain. 2014. PMID: 24277719
-
Prenatal Zinc Deficient Mice as a Model for Autism Spectrum Disorders.Int J Mol Sci. 2022 May 29;23(11):6082. doi: 10.3390/ijms23116082. Int J Mol Sci. 2022. PMID: 35682762 Free PMC article. Review.
-
No effect of prenatal vitamin D deficiency on autism-relevant behaviours in multiple inbred strains of mice.Behav Brain Res. 2018 Aug 1;348:42-52. doi: 10.1016/j.bbr.2018.04.004. Epub 2018 Apr 12. Behav Brain Res. 2018. PMID: 29655594
-
Loss of COMMD1 and copper overload disrupt zinc homeostasis and influence an autism-associated pathway at glutamatergic synapses.Biometals. 2014 Aug;27(4):715-30. doi: 10.1007/s10534-014-9764-1. Epub 2014 Jul 10. Biometals. 2014. PMID: 25007851
-
Ultrasonic vocalizations in Shank mouse models for autism spectrum disorders: detailed spectrographic analyses and developmental profiles.Neurosci Biobehav Rev. 2014 Jun;43:199-212. doi: 10.1016/j.neubiorev.2014.03.021. Epub 2014 Apr 12. Neurosci Biobehav Rev. 2014. PMID: 24726578 Review.
Cited by
-
Shank2/3 double knockout-based screening of cortical subregions links the retrosplenial area to the loss of social memory in autism spectrum disorders.Mol Psychiatry. 2022 Dec;27(12):4994-5006. doi: 10.1038/s41380-022-01756-8. Epub 2022 Sep 13. Mol Psychiatry. 2022. PMID: 36100669 Free PMC article.
-
Zinc is a key regulator of gastrointestinal development, microbiota composition and inflammation with relevance for autism spectrum disorders.Cell Mol Life Sci. 2021 Dec 22;79(1):46. doi: 10.1007/s00018-021-04052-w. Cell Mol Life Sci. 2021. PMID: 34936034 Free PMC article.
-
Characterization of zinc amino acid complexes for zinc delivery in vitro using Caco-2 cells and enterocytes from hiPSC.Biometals. 2017 Oct;30(5):643-661. doi: 10.1007/s10534-017-0033-y. Epub 2017 Jul 17. Biometals. 2017. PMID: 28717982 Free PMC article.
-
Lack of Vesicular Zinc Does Not Affect the Behavioral Phenotype of Polyinosinic:Polycytidylic Acid-Induced Maternal Immune Activation Mice.Front Behav Neurosci. 2022 Feb 22;16:769322. doi: 10.3389/fnbeh.2022.769322. eCollection 2022. Front Behav Neurosci. 2022. PMID: 35273483 Free PMC article.
-
Sex-specific Behavioral Features of Rodent Models of Autism Spectrum Disorder.Exp Neurobiol. 2018 Oct;27(5):321-343. doi: 10.5607/en.2018.27.5.321. Epub 2018 Oct 31. Exp Neurobiol. 2018. PMID: 30429643 Free PMC article. Review.
References
-
- CDC (Centers for Disease Control Prevention) (2014). Prevalence of autism spectrum disorder among children aged 8 years - autism and developmental disabilities monitoring network, 11 sites, United States, 2010. MMWR Surveill. Summ. 63, 1–21. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

