Experimental and Computational Evidence for a Loose Transition State in Phosphoroimidazolide Hydrolysis
- PMID: 26974265
- PMCID: PMC7547882
- DOI: 10.1021/jacs.6b00784
Experimental and Computational Evidence for a Loose Transition State in Phosphoroimidazolide Hydrolysis
Abstract
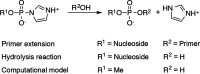
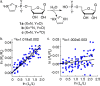
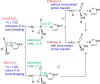
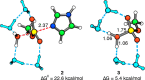
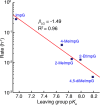
Phosphoroimidazolides play a critical role in several enzymatic phosphoryl transfer reactions and have been studied extensively as activated monomers for nonenzymatic nucleic acid replication, but the detailed mechanisms of these phosphoryl transfer reactions remain elusive. Some aspects of the mechanism can be deduced by studying the hydrolysis reaction, a simpler system that is amenable to a thorough mechanistic treatment. Here we characterize the transition state of phosphoroimidazolide hydrolysis by kinetic isotope effect (KIE) and linear free energy relationship (LFER) measurements, and theoretical calculations. The KIE and LFER observations are best explained by calculated loose transition structures with extensive scissile bond cleavage. These three-dimensional models of the transition state provide the basis for future mechanistic investigations of phosphoroimidazolide reactions.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
- Inoue T.; Orgel L. E. J. Am. Chem. Soc. 1981, 103, 7666.10.1021/ja00415a051. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

