Cardiomyocytes in Young Infants With Congenital Heart Disease: a Three-Month Window of Proliferation
- PMID: 26976548
- PMCID: PMC4791641
- DOI: 10.1038/srep23188
Cardiomyocytes in Young Infants With Congenital Heart Disease: a Three-Month Window of Proliferation
Abstract
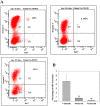
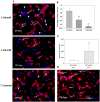
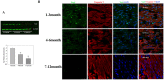
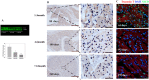
Perinatal reduction in cardiomyocyte cell cycle activity is well established in animal models and humans. However, cardiomyocyte cell cycle activity in infants with congenital heart disease (CHD) is unknown, and may provide important information to improve treatment. Human right atrial specimens were obtained from infants during routine surgery to repair ventricular septal defects. The specimens were divided into three groups: group A (age 1-3 months); group B (age, 4-6 months); and group C (age 7-12 months). A dramatic fall in the number of Ki67-positive CHD cardiac myocytes occurred after three months. When cultured in vitro, young CHD myocytes (≤ 3 months) showed more abundant Ki67-positive cardiomyocytes and greater incorporation of EdU, indicating enhanced proliferation. YAP1 and NICD-important transcript factors in cardiomyocyte development and proliferation-decreased with age and β-catenin increased with age. Compared with those of older infants, cardiomyocytes of young CHD infants (≤ 3 months) have a higher proliferating capacity in vivo and in vitro. From the perspective of cardiac muscle regeneration, CHD treatment at a younger age (≤ 3 months) may be more optimal.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

