Role of monocytes and macrophages in regulating immune response following lung transplantation
- PMID: 26977996
- PMCID: PMC4858348
- DOI: 10.1097/MOT.0000000000000313
Role of monocytes and macrophages in regulating immune response following lung transplantation
Abstract
Purpose of review: Advances in the field of monocyte and macrophage biology have dramatically changed our understanding of their role during homeostasis and inflammation. Here we review the role of these important innate immune effectors in the lung during inflammatory challenges including lung transplantation.
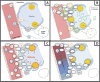
Recent findings: Neutrophil extravasation into lung tissue and the alveolar space have been shown to be pathogenic during acute lung injury as well as primary graft dysfunction following lung transplantation. Recent advances in lung immunology have demonstrated the remarkable plasticity of both monocytes and macrophages and demonstrated their importance as mediators of neutrophil recruitment and transendothelial migration during inflammation.
Summary: Monocytes and macrophages are emerging as key players in mediating both the pathogen response and sterile lung inflammation, including that arising from barotrauma and ischemia-reperfusion injury. Ongoing studies will establish the mechanisms by which these monocytes and macrophages initiate a variety of immune response that lay the fundamental basis of injury response in the lung.
Conflict of interest statement
Conflicts of interest: None.
Figures
References
-
- Mitchell AJ, Roediger B, Weninger W. Monocyte homeostasis and the plasticity of inflammatory monocytes. Cellular immunology. 2014;291(1–2):22–31. In this comprehensive review the authors discuss the monocyte development and homeostatic control of monocyte populations. - PubMed
-
- Serbina NV, Pamer EG. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nature immunology. 2006;7(3):311–7. - PubMed
-
- Landsman L, Varol C, Jung S. Distinct differentiation potential of blood monocyte subsets in the lung. Journal of immunology. 2007;178(4):2000–7. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

